The Flint Water Crisis: What's Really Going On?
By Adrian Dingle December 2016
When Flint’s water started smelling foul, tasting odd, turning orange and the children drinking it started to develop nasty skin rashes, there was clearly something terribly wrong. But what? That was a mystery that only chemistry could solve.
In the spring of 2014, large amounts of lead found their way into the water that some residents of Flint, Mich., were using to drink, cook, and brush their teeth. Lead is known to be particularly damaging to young children’s developing brains. So how did lead get into the tap water in Flint?
Treating and supplying water
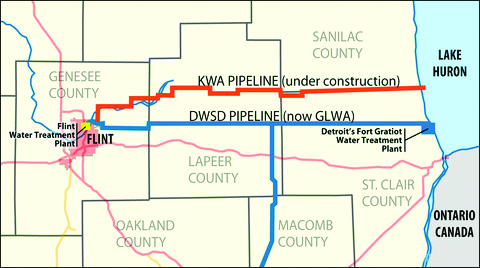
In early 2013, in order to save money, Flint officials decided to change the source of the city’s water from the Detroit Water and Sewerage Department to the Karegnondi Water Authority (KWA). However, KWA was not ready to deliver the water, because they were in the process of building a new pipeline to bring water from nearby Lake Huron. This meant that officials in the city of Flint needed to find a short-term solution while the new pipeline was being built. They decided to use water from the local Flint River that they would clean in water treatment plants.
In April 2014, Flint switched over to the new source, and the problems began almost immediately. Within weeks, residents were complaining of foul-smelling and discolored water. The city initially denied that there were any issues, but within a few months, they issued a boil-water advisory. Later, in October 2014, the General Motors car plant in Flint announced that they would no longer use the water, because they feared it would cause corrosion within the factory.
A few months later, Flint told its residents that their water had high levels of organic molecules called trihalomethanes. These molecules are similar to methane molecules (CH4), but three of the hydrogen atoms are replaced with halogen (group 17) atoms—fluorine, chlorine, bromine, or iodine. The four most common trihalomethanes are dibromochloromethane (CHClBr2), dichlorobromomethane (CHCl2Br), trichloromethane (CHCl3)—better known as chloroform—and tribromomethane (CHBr3).
Trihalomethanes are a concern because they have been linked with numerous health concerns, including liver, kidney, lung, and heart problems. When the chorine used to disinfect water reacts with algae, leaves, and weeds, trihalomethanes are produced.
Toxic levels of lead
Trihalomethanes are just one group of chemicals associated with water contamination, lead is another. Lead was a popular choice for use as water pipes for centuries. The Romans used the dense metal because of its durability and malleability. Water pipes are no longer made from lead, but older cities, such as Flint, still rely on lead pipes—in addition to those made from copper and iron—to transport water to people’s homes. No one knows exactly how many lead pipes are used in the United States, but the number is in the millions. In addition to the pipes themselves, lead is also found in alloys (mixtures of metals) in other parts of the plumbing system. For example, the solder used to join pipes together is an alloy of tin and lead.
In Flint, massive levels of lead entered the water system. In one sample of water taken from a Flint home, a lead level of 13,000 parts per billion (ppb) was found. The chemists who tested the water were skeptical at first. But after repeating the experiment a number of times, they found that the 13,000 ppb reading was accurate.
It is important to note that no level of lead in the water supply is considered safe, but the U.S. Environmental Protection Agency (EPA) has set a maximum level of lead contamination in the tap water in 90% of homes at 15 ppb—also known as the “action level.” That particular sample of Flint water had a lead level close to 1,000 times the action level.
Lead-prevention techniques
Oxidizing agents in water such as dissolved oxygen can react with exposed lead pipes causing soluble Pb2+ ions to enter the supply. Two chemical reactions are involved: 1) oxidation, and 2) reduction. Both reactions are called redox reactions, for short. In the oxidation reaction, lead loses electrons and forms Pb2+ ions, as follows:
2 Pb(s) → 2 Pb2+(aq) + 4 e–
In the reduction reaction, oxygen gains electrons, as follows:
4 e– + 2 H2O (l) + O2 (aq) → 4 OH– (aq)
Together, the oxidation and reduction reactions combine to give the full redox reaction:
2 Pb (s) + O2 (g) + 2 H2O (l) → 2 Pb(OH)2 (s)
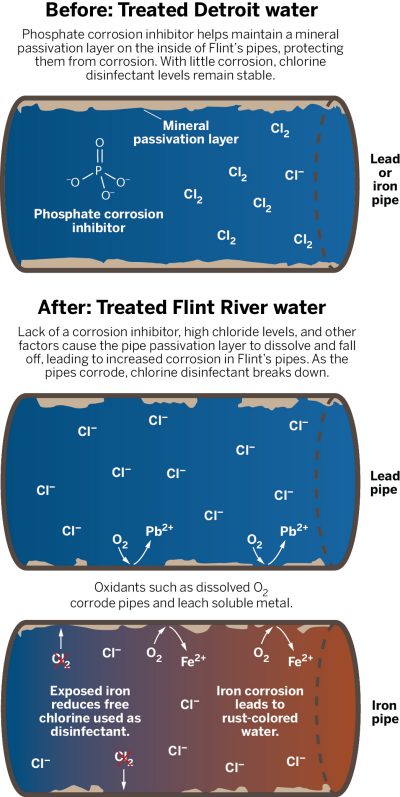
Cities use various methods to prevent lead pipes from leaching lead ions into the water supply. One method is to create a protective layer on the inside of the pipe. This is done by adding phosphate ions (PO43–) to the water supply. Phosphate is also known as orthophosphate. These negative polyatomic ions help to create lead(II) phosphate (Pb3(PO4)2), an insoluble, mineral-like crust on the inside of the pipes.
The addition of phosphate ions to the water needs to be an ongoing, continual process, and if the crust is maintained over time, the water comes into less or no contact with the lead pipe itself and, as a result, less redox reactions take place and less lead enters the water. If the phosphate crust is not maintained, it breaks down over time and lead can leach into the water supply.
The water that Flint used to get from nearby Detroit was treated with phosphate ions in this way and, as a result, remained relatively free of lead ions. When Flint changed its water supply to river water, authorities failed to treat it with phosphate ions, so the phosphate crust broke down over time and lead ions entered the water.
Complicating factors
In addition to the loss of the phosphate protective layer in the pipes, two other problems occurred, as well.
First, the Flint River water had some unusually high levels of chloride ions, which can accelerate the corrosion of the pipes. In part, these high chloride levels came from salts used to treat roads during the cold and snowy Michigan winters. Often, chlorides enter rivers as run-off from the roads. This is actually an example of connections between different applications of chemistry: De-icing Michigan’s slippery roads has some immediate and obvious benefits, but the run-off of chloride ions can be an unintended consequence.
Second, the pH of the water from the Flint water treatment plants was too low. By maintaining a relatively high pH (around 10), the solubility of another lead compound that contributes to the protective layer, lead(II) carbonate (PbCO3), decreases. This helps to maintain the protective layer on the pipes. If the pH is too low, then the protective layer may dissolve and fall off. We can explain this by considering Le Châtelier’s principle, which states that a system that is shifted away from equilibrium acts to restore the equilibrium by reacting in opposition to the shift.
Lead(II) carbonate is relatively insoluble in water, but does dissolve a little, and in the process sets up this equilibrium:
PbCO3 (s) ⇌ Pb2+ (aq) + CO32– (aq)
The carbonate ions that are produced can then react with any hydrogen ions (acid) present in the water supply, according to this equation:
CO32– (aq) + H+ (aq) → H2O (l) + CO2 (g)
The lower the pH, the more hydrogen ions that are present in solution and the more the carbonate ions react. The removal of carbonate ions from the equilibrium system of lead(II) carbonate in the first equation results in an increase in the concentration of lead ions. To restore the equilibrium, the chemical reaction shifts to the right, so that additional carbonate ions (and lead ions) are produced.
To ensure that the number of lead and carbonate ions is the same on the right side of the equation, the reaction then shifts to the left and to the right a few times until a new equilibrium is reached. In this new equilibrium, the number of lead ions is the same as the number of carbonate ions, but the number of lead ions is larger than what it was during the original equilibrium (before the carbonate ions were removed), resulting in more lead ions at equilibrium than before.
Flint’s water was found to have pH values between 7 and 8, which is not basic enough to prevent lead carbonate from dissolving. The lack of a protective layer in the iron pipes can cause a similar oxidation reaction to the one occurring in the lead pipes:
Fe(s) → Fe2+ (aq) + 2 e–
The aqueous iron ions can cause the water to turn an unsightly rust color, but it can also have another troublesome side effect. This time, elemental chlorine (Cl2) can be reduced to chloride ions (Cl–). When the electrons are given up by the iron and picked up by the chlorine molecules, the following reduction reaction occurs:
2 e– + Cl2(aq) → 2 Cl–(aq)
The loss of elemental chlorine is a potentially huge problem for public health. Chlorine is added to the water supply to eliminate water-borne pathogens. The removal of elemental chlorine via reduction can mean that water-borne pathogens have a better chance of surviving, causing disease.
How is Flint’s water now?
In October 2015, Flint switched back to the Detroit system as the source of its water. But this incident will still have some far-reaching consequences.
At the top of the list are the potential ongoing health problems that the estimated 6,000 to 12,000 children may have to cope with. Nobody can predict what those problems may be, but high levels of lead exposure have been linked to developmental and behavioral problems in children and issues related to brain function. It will likely be a long time into the future before all of the problems come to light, but the concerns are real and troubling for all of the families affected.
In addition to the long-term health concerns, a number of lawsuits have been filed against Flint and Michigan officials, and millions of dollars will be required to address the crisis and rebuild the water-pipe system. In January 2016, the U.S. government pledged $80 million in aid to the state of Michigan, most of which is to be used to repair the infrastructure of Flint’s water-supply system. In February of the same year, the U.S. Department of Health and Human Services announced that $500,000 would be made available to two Flint area health centers to help fight the problems caused by the tainted water.
Through knowledge of chemistry, including the formation of precipitates, the pH scale, and the redox chemistry of half-reactions, scientists were able to solve the Flint water mystery. The crisis was stopped before it became worse, and Flint’s water supply was made safe—all thanks to chemistry.
Selected references
How Lead Ended Up In Flint’s Tap Water. Chem. & Eng. News, 94 (7), Feb 11, 2016, pp 26–29.
Haines, G. K. Is This Water Recycled Sewage? ChemMatters, Feb 2011, pp 8–10.
Walters, L. A. Flint Water Study Updates. Virginia Polytechnic Institute and State University: http://flintwaterstudy.org/ [accessed June 2016].
Dennis, B. Water in Flint Is Improving but Still Unsafe to Drink, Researchers Say. The Washington Post, April 12, 2016: http://tinyurl.com/zdn3o2a [accessed June 2016].
Adrian Dingle is a science writer who lives in Atlanta, Ga. This is his first article in ChemMatters.
Timeline of Events

April 2013: Facing severe financial pressures, the city of Flint, Mich., decides to change the source of the city's water and signs a contract with the Karegnondi Water Authority (KWA), but KWA would not supply water until 2016.

April 2014: Flint begins using water from the Flint river until the KWA pipeline from Lake Huron is completed.
May 2014: Flint residents begin to complain about the water.

August 2014: Water tests positive for E. coli bacteria; a boil-advisory is issues in parts of Flint.

January 2015: Flint announces that the water contains high levels of trihalomethanes, in violation of the Safe Drinking Water Act, but city officials tell residents that the water is safe.

February 2015: Flint resident LeAnne Walters’s son develops rashes. High levels of lead are detected in the water in her home.

September 2015: Data show that many children have elevated lead levels in their blood.

October 2015: Flint switches back to using water from Detroit.

January 2016: Michigan Gov. Rick Snyder declares a state of emergency in Flint. Then, President Barack Obama signs an emergency declaration and orders federal aid for Flint.

January 2016: Volunteers from around the country, including students from Harper Woods High School, Harper Woods, Mich., donate water and supplies to Flint residents.

March 2016: The Flint water task force, appointed by Governor Snyder, finds that state agencies within Michigan are mainly responsible for the crisis.
Not Just in Flint
The Flint water crisis may be the most serious lead contamination in drinking water to date, but other water supplies across the United States have been affected, too. In 2001, Washington, D.C. changed how it treated drinking water, and lead levels dramatically rose. In 2006, unsafe levels of lead were found in Durham and Greenville, N.C. In the summer of 2015, officials discovered significant lead contamination in drinking water in Jackson, Miss., but waited six months before informing residents. The list goes on.
The U.S. Congress banned the use of lead water pipes 30 years ago, but the rule applies only to new water pipes. Millions of older ones remain in use. One possible solution is to dig up and replace these lead pipes, as has occurred between 2001 and 2011 in Madison, Wis. But doing so is costly, and not all cities have the financial means to do it. Other solutions include adding more or different chemicals to the source water and to continuously test for contaminants. The problem is not easy to solve but at least the Flint water crisis has raised awareness of the issue, and it may lead to safer drinking water throughout the United States in the future.
—Adrian Dingle