Separation of Rare Earth Elements by Charles James
Dedicated October 29, 1999, at the University of New Hampshire in Durham, New Hampshire.
Charles James, a chemistry professor at the University of New Hampshire from 1906 to 1928, was an internationally-recognized expert in rare earth chemistry. In a laboratory in Conant Hall, James devised novel fractional crystallization techniques for separating rare earth elements, which were widely adopted by other chemists. James used his method to separate large amounts of ytterbium, previously considered to be a single element, into two elements now known as ytterbium and lutetium.
Contents
Charles James and the Rare Earths
The 17 rare earth elements, or lanthanides, consist of elements 21 (scandium), 39 (yttrium), and 57 (lanthanum) to 71 (lutetium). The rare earths were so named because of their low concentration in minerals which were scarce. However, some of the elements are not as rare as once thought. Today, the rare earths are important for their use in hard drives and wind turbine generators, glass polishes, ceramic glazes, protective goggles, lasers and superconductors. More recently, some have been used in diagnostic imaging in the field of nuclear medicine.
A University of New Hampshire (UNH) professor, Charles James (1880-1928), all but forgotten today, was well known and highly respected by his contemporaries for his contribution to rare earth chemistry. The very first paper published by James in the Journal of the American Chemical Society, "A New Method for the Separation of the Yttrium Earths," indicates the direction of his research. Keenly motivated to improve the methods of separation and endowed with imagination and determination, James experimented with the preparation of many different salts of the rare earths derived from simple inorganic to complex organic acids. Letters in the UNH archives show that he supplied numerous workers worldwide with samples of the compounds that he had prepared, including the sample of terbium that Henry Moseley, an English scientist, used to determine its atomic number. James's use of bromates and double magnesium nitrates for fractional crystallization became known as the "James Method." Developed in the early 1900s, his method was used widely by others and remained the best for separating rare earth elements until the advent of ion exchange in the 1940s.
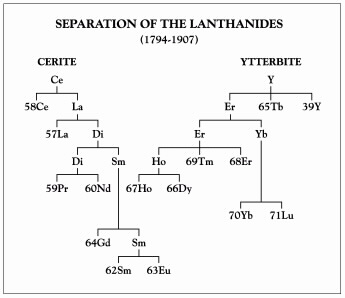
James's work with the rare earths is part of a long history that illuminates more areas of chemical progress than does any other group of elements. It stretches from the discovery of the first rare earth mineral in 1787 by Carl Axel Arrhenius to the unequivocal identification of the last in 1947 by Jacob Marinsky, Lawrence Glendenin and Charles Coryell. The story involves the discovery of emission spectroscopy by Gustav Robert Kirchhoff and Robert Wilhelm Bunsen, the development of the periodic table by Dmitri Ivanovich Mendeleev and Lothar Meyer, and the discovery of X-rays by Wilhelm Conrad Roentgen. It includes the formulation of atomic numbers by Moseley, atomic structure by Neils Bohr and Ernest Rutherford, and work on the atomic bomb during World War II. Over the course of a century, two minerals — ytterbite and cerite — were separated into eight and seven stable elements, respectively, as shown in the diagram at right.
Discovery of the Rare Earth Elements
The story of their discovery is probably the most confusing and complex of any of the elements. The search for and identification of the rare earth elements constituted an integral part of the development of science and technology in the late 19th and early 20th centuries. Several factors made their identification difficult: The chemical and physical properties of the different elements are quite similar; the elements were isolated as "earths" or oxides of the elements; and the methods of separation and purification depended on laborious fractional precipitation and crystallization techniques. Compounding the difficulties of separation were a lack of good methods for identification and assessment of purity and a lack of knowledge of the number of rare earth elements that existed.
The first rare earth element, yttrium, was isolated in 1794 by the Swedish chemist Johan Gadolin from a heavy black mineral, ytterbite, named for the village of Ytterby where it was found. In 1803, Jons Jacob Berzelius and Wilhelm Hisinger in Sweden and Martin Klaproth in Germany announced almost simultaneously that they had isolated a new element, cerium, from the mineral cerite. This provoked the first of several priority disputes in the pathway to the discovery of rare earth elements.
The invention of the spectroscope by Kirchhoff and Bunsen in 1859 and the development of spectral analysis, along with the development of the periodic table by Mendeleev and Meyer in 1869, provided valuable tools for the study of rare earths. The impact of these advances is clear. In the 60 or so years before their introduction, only six rare earths had been identified, one of them erroneously. In the following 50 years, the number jumped to 15. However, problems remained. The rare earths severely challenged Mendeleev's periodic system, because he did not know where to place them in the table. He made many modifications to accommodate them. Meanwhile, spectroscopic analysis was causing difficulties.
Because of the complexity of the spectra of the rare earths and the questionable purity of the samples, many claims were being made for new elements that proved false. Despite these difficulties, by 1901 only two rare earths were still undiscovered. In 1907, almost simultaneously, Georges Urbain in France, Carl Auer von Welsbach in Austria and Charles James in the United States separated ytterbium into two fractions. Urbain's designation of the new element as lutetium, after the Roman name for his native city, Paris, was accepted by most, although von Welsbach's name cassiopium was used in German-speaking countries for many years. Von Welsbach contested Urbain's priority for the discovery of lutetium. James, although he had accumulated a substantial amount of highly purified lutetium oxide, withdrew a paper he had submitted for publication and made no public claim for his own work.
Lutetium was the 16th of the rare earths to be identified. Using X-ray spectroscopy, Moseley demonstrated that only one rare earth element remained to be discovered: element 61. He determined the atomic numbers of the elements and showed that they were arranged in the periodic table in order of atomic number, not atomic weight, as Mendeleev had thought. In the 1920s, James, B. Smith Hopkins and Luigi Rolla each thought he had isolated element 61. Hopkins's publication preceded that of James, and so Hopkins's designation of the element as illinium was accepted temporarily. The last of the priority disputes over discovery of a rare earth element ended inconclusively, however, when none of the claims could be substantiated. The authentic discovery of element 61 had to await the development of a new separation technique, ion-exchange chromatography, and work on the atomic bomb during World War II. Finally, in 1947, Marinsky, Glendenin and Coryell announced the discovery of the unstable element 61, which they named promethium after the Titan who stole fire from the gods.
Chemistry at the University of New Hampshire, 1866-1928
Few contributions to the understanding of the rare earths could overshadow the achievements accomplished in the chemistry laboratories at UNH. Chemistry has been a part of the university from its beginning. UNH traces its origins to the acceptance of the Morrill Act by the New Hampshire Legislature in June 1866, which led to the establishment of the New Hampshire College of Agriculture and Mechanic Arts in Hanover, in association with Dartmouth College. The first faculty member of the college in 1868 was Ezekiel W. Dimond, who was appointed professor of general and applied chemistry. An 1865 graduate of Middlebury College, Dimond was synonymous with the institution in its early days. Not only was he professor of chemistry, he was also the president, business manager, architect, supervisor of construction and lobbyist in the state legislature. His efforts led to the construction of Culver Hall, which contained a chemistry laboratory, described in 1874 as the only one in the state. Thus the Department of Chemistry is the only department that has existed for the entire history of the University. Dimond served as professor of chemistry until his untimely death, at the age of 40, in 1876.
Succeeding Dimond as head of chemistry were Benjamin T. Blanpied (1877-1889), Fred W. Morse (1889-1909) and Charles L. Parsons (1909-1911). When Parsons resigned in 1911 to become chief chemist of the U.S. Bureau of Mines, Charles James succeeded him and served as professor and head of the Department of Chemistry from 1912 until his death in 1928. For the most part, the department focused on the inorganic and analytical disciplines under his leadership.
While the New Hampshire College of Agriculture and Mechanic Arts was still located on the campus of Dartmouth College at Hanover, the State of New Hampshire was bequeathed the entire fortune of a Durham farmer, Benjamin Thompson. He specified that an agricultural college be founded on his property in Durham. In 1891, preparations began for the college to move to Durham. Charles Parsons designed the new chemistry laboratories in Conant Hall, which was constructed during 1892-1893 at a cost of $40,000 to house the Department of Chemistry and other science departments. From 1906 to 1928, Conant Hall also housed the offices and laboratories of James.
By the late 1920s, Conant Hall was no longer adequate to accommodate chemistry; James designed a new building for chemistry and agricultural chemistry. Completed after James's death, the Charles James Hall was dedicated at the 1929 fall meeting of the Northeastern Section of the American Chemical Society (ACS). Conant Hall is now one of the three surviving original buildings of UNH at Durham. The interior has been completely altered since its use by the Department of Chemistry, and today it serves as a classroom and office building. The exterior, however, is remarkably unchanged from its appearance in 1893, when the Department of Chemistry first occupied Conant Hall and when, in subsequent years, James and his students conducted the thousands of manipulations needed to complete rare earth separations. Conant Hall and Durham became known worldwide among chemists as a center of rare earth research.
Biography of the Chemist Charles James
Charles James was born in Earls Barton, near Northampton, England, on April 27, 1880. He received his primary and secondary education in the nearby town of Wellingborough. At the age of 15, his interest in chemistry was kindled by the discovery of several of his father's old chemistry books, and he set up a laboratory in the family home.
In 1899, he entered the Institute of Chemistry, University College, London, where he studied under Sir William Ramsay and John Norman Collie. In 1900, he was awarded the Ramsay Silver Medal for his work. Ramsay wrote, "James is certainly able to take his place with any of the [American chemists], allowing for his age, in originality and power for work. He was always original ever since he came as a youngster to my laboratory; he did not tire of his work in our college, but carried out work at his home in Kettering, about 50 miles from London, where he had a small laboratory of his own." He passed the examinations for associate in 1904 and for fellow in 1907 at the Institute of Chemistry. He also supplemented his study of chemistry with industrial experience at the New Carnsley Iron and Steel Co. in Kettering, England.
In 1906, he accepted a position at the National Refining Company in Westchester, New York, and in February of that year, he was called to the New Hampshire College of Agriculture and Mechanic Arts by Charles Parsons to become assistant professor of chemistry.
While in Durham, James conducted his extensive investigations of the chemistry of the rare earths. The results were described in more than 60 papers published primarily in the Journal of the American Chemical Society between 1907 and 1931. He separated and characterized large quantities of pure rare earths, frequently at the kilogram level. He discovered new compounds of neodymium, samarium and europium and determined the atomic weights of samarium, thulium and yttrium. More than 200 specimens of his rare earth compounds still remain in the James Collection at UNH.
His students and contemporaries knew James as a dedicated and meticulous chemist and a sympathetic mentor. Much of his rare earth work was carried out with the help of undergraduates whom he patiently developed into accomplished researchers. He was known by all as "King" James, a tribute to his English heritage.
The New York Section of ACS awarded James the Nichols Medal in 1911 for his research in the chemistry of the rare earth elements. In 1927, James received an honorary doctor of science degree from UNH.
In 1915, he married Marion E. Templeton, a member of the faculty of the Department of Botany. Their common interest in floriculture made their residence with its wide variety of flowers a showplace of Durham. He was especially interested in the propagation of rare plants, which he collected, along with rare minerals, during summer vacations. James died on Dec. 10, 1928.
Further Reading
- The Life and Work of Charles James (University of New Hampshire Magazine)
Landmark Designation and Acknowledgments
Landmark Designation
The American Chemical Society designated the separation of rare earth elements by Charles James as a National Historic Chemical Landmark at the University of New Hampshire on October 29, 1999. The plaque commemorating the event reads:
Beginning in 1906, in a laboratory in Conant Hall, Charles James (1880-1928) devised novel fractional crystallization techniques for separating rare earth elements, which were widely adopted by other chemists. James used his method to separate large amounts of ytterbium, hitherto considered to be a single element, into two elements now known as ytterbium and lutetium. When the simultaneous isolation of lutetium was published in 1907 by Georges Urbain, James made no public claim for his own pioneering work. Despite his retiring nature, James was internationally recognized as an expert in rare earth chemistry. His highly purified rare earth specimens were in demand by research laboratories throughout the world.
Acknowledgments
Adapted for the internet from “Separation of Rare Earth Elements,” produced by the National Historic Chemical Landmarks program of the American Chemical Society in 1999.
Back to National Historic Chemical Landmarks Main Page.
Learn more: About the Landmarks Program.
Take action: Nominate a Landmark and Contact the NHCL Coordinator.




