Discovery of Highly Active Antiretroviral Therapy for HIV
Dedicated at Merck in West Point, Pennsylvania, on Oct. 7, 2022, and GSK in Collegeville, Pennsylvania on May 15, 2024
On June 5, 1981, national health experts published accounts of a strange infection circulating around Los Angeles. The U.S. Centers for Disease Control and Prevention (CDC) reported on that day that, within the previous eight months, five men showed up at area hospitals with rare pneumonias. The men didn’t know each other. They had no common contacts. They were young and previously healthy. Yet, here they were, gravely ill with a type of pneumonia only seen in people with severely suppressed immune systems. Two died before the report was published. All were described as members of the gay community.
By July of 1981, the CDC had uncovered many more cases of rare diseases that took advantage of people’s weakened immune systems, such as Kaposi’s sarcoma. Experts raced to appraise the mysterious outbreak. Hundreds received diagnoses and many died within a year. By September 1982, the epidemic was named acquired immune deficiency syndrome (AIDS). It took one more year for scientists to find the pathogen responsible for AIDS: human immunodeficiency virus (HIV).
Over 100,000 people died of AIDS in the U.S. in the 1980s. Doctors quickly learned that HIV infection — which spreads through sexual transmission and other routes — was not limited to specific population groups; it affected people regardless of their sexual orientation and gender identity. One weapon wouldn’t be enough to fight it: Although the first HIV drug earned approval in 1987, annual deaths continued to climb. Pharmaceutical researchers experimented with ways to attack the disease from multiple angles. Over the next decade, they developed drugs that work synergistically to defend people from the infection. The result of this research, highly active anti-retroviral therapy, or HAART, has saved millions of lives and turned a once-seemingly undefeatable pandemic into a manageable chronic disease.
Combating HIV, through development of HAART, took prodigious interdisciplinary efforts. Researchers brought scientific ingenuity to each step — thinking on the fly while people’s lives hung in the balance, and following every sliver of hope they could find.
Contents
Without HIV treatment, people with AIDS typically survive about three years.”
— About HIV, CDC
The retrovirus
HIV infection is a master class in destruction; human immune systems are essentially defenseless against it. So to even the odds, drug developers sought to understand each step of the takeover of the immune system. Among the questions they asked: How does HIV enter a human cell? And how does it force that cell to carry out its deadly plans?
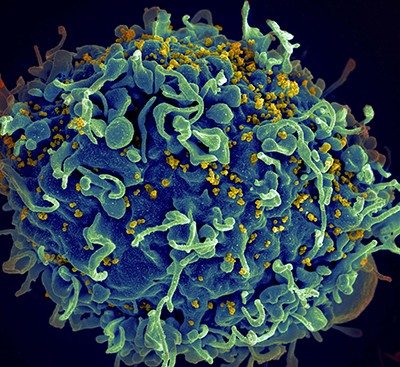
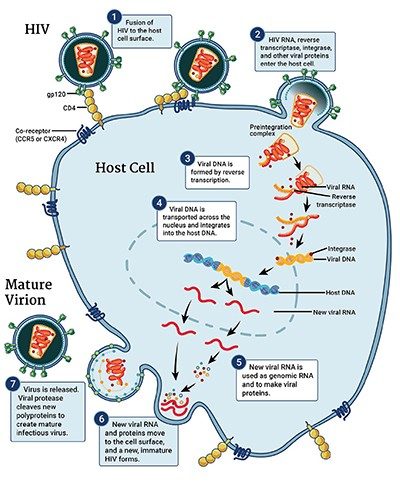
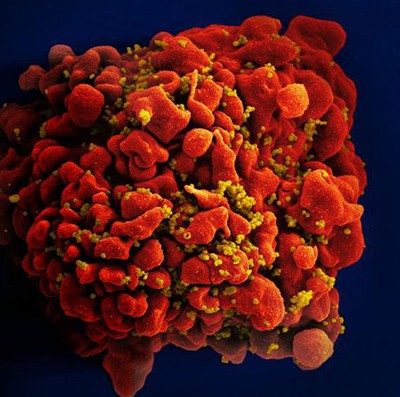
HIV is a spherical virus. It contains proteins and two copies of the genetic material known as RNA within its shell. Dotting the surface of that shell are molecules that can recognize CD4+ T cells, which are human immune cells. When HIV comes into contact with CD4+ T cells, the virus fuses with and delivers its contents into the human cells.
Inside the cells, the virus makes copies of itself. Usually, cells turn DNA into RNA — but the opposite happens with this virus, and so HIV is known as a “retrovirus.” An enzyme attached to HIV’s RNA, called reverse transcriptase, converts the virus’s RNA template into DNA. It assembles genetic building blocks into a single strand of DNA. From that single strand, the reverse transcriptase enzyme repeats the process to complete a segment of double-stranded DNA.
Another enzyme, called integrase, tows the genetic material to the person’s own DNA and integrates the interloper into the sequence. The new double-stranded DNA segment looks like a fragment of a person’s normal genetic blueprint, but in this case, it’s a blueprint for the human cell to follow the virus’s instructions. That leads to the production of more HIV RNA.
Some RNA goes on to create proteins for the scaffold of new HIV virus particles; other copies hide away until the new particles infect other cells. Without any drug to stop it, HIV copies itself relentlessly in human immune cells — thousands of times inside each one until the cell dies. HIV levels rise. Immune cell counts plummet. And an infected person’s health deteriorates as they become less able to fight off other infections.
What is HIV?
HIV (human immunodeficiency virus) is a virus that attacks the body’s immune system. If HIV is not treated, it can lead to AIDS (acquired immunodeficiency syndrome). There is currently no effective cure. Once people get HIV, they have it for life. But with proper medical care, HIV can be controlled. People with HIV who receive effective HIV treatment can live long, healthy lives and protect their partners. (Source: CDC)
View larger image
A drug finds new purpose
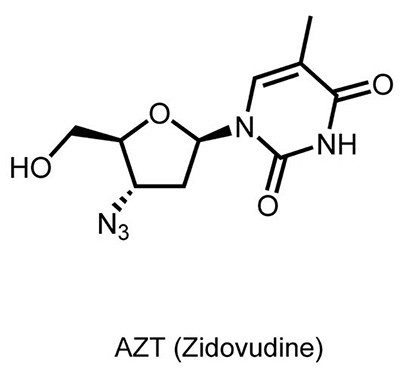
The first HIV drug, zidovudine (also known as Retrovir®, azidothymidine, or AZT), was developed decades before the first known case of HIV. Originally developed to treat cancer, it was a failure against that disease.
In the 1960s, medicinal chemists suspected that viruses cause most cancers, so they searched for drugs to interrupt viral multiplication of genetic material. A team of researchers from the Wayne State University School of Medicine set their sights on the reverse transcriptase enzyme, which turns viral RNA into DNA. They created AZT, hoping it would stop reverse transcriptase — thereby stopping a virus, and stopping cancer. But it didn’t work. They abandoned AZT research in 1967 after it failed to treat one type of leukemia in mice.
Seven years later, scientists in Germany proved that AZT works against the Friend virus, a retrovirus that causes a different type of leukemia in rodents. While this gave concrete evidence that AZT fights retroviruses, there was still no evidence that this mattered for human diseases.
Then, in the 1980s, HIV thundered into the public health world. In 1984, researchers in France and the U.S. established that a retrovirus causes AIDS. The following year, Abbott Laboratories developed the first blood screen for the virus.
Experts rejoiced when AZT appeared potent against HIV, in Petri dish experiments and animals. Clinical trials in humans began in 1985; approval by the U.S. Food and Drug Administration (FDA) came in 1987. AZT became the world’s first antiretroviral therapy, targeting one key enzyme in HIV’s cellular takeover strategy. The treatment was developed and marketed by Burroughs Wellcome, which became part of the firm now known as GSK.
In 1985… we didn’t have any treatment. People didn’t make it out of the hospital.”
— Judith Currier, University of California, Los Angeles, 2012, The Plague of Our Time, documentary by New England Journal of Medicine
'Highly active'
AZT helped address the AIDS crisis in the late 1980s, but disabling just one step of HIV’s takeover tactics didn’t work as well as experts hoped. When taken in high doses, the drug had toxic side effects. And the virus could adapt quickly to it, especially when people missed a dose. By the mid-1990s, it was clear from clinical trials that the benefit of AZT therapy lasted only about a year.
Meanwhile, researchers from Merck and elsewhere had begun to unravel the importance of another bit of HIV’s biological machinery: the protease enzyme. After HIV infects a host cell, ribosomes read genetic blueprints on its viral messenger RNA, and use those instructions to churn out long protein chains. Those protein chains aren’t active as is, but HIV protease chops them into smaller subunits, which are active.
Researchers at Merck, and shortly thereafter at Roche, described this role of HIV protease in HIV infections in 1988. They found that snipping proteins is critical for the virus to replicate. Now, they just needed to design a drug that could target and prevent that step.
Their goal was to design a drug to interfere with the function of the protease, something that would jam into the protease’s structure and prevent it from snipping those long protein chains. Finding the right molecule to satisfy all of a drug’s requirements takes years of basic chemistry, computer modeling, and trial and error. Researchers had to consider factors including:
- The size and shape of the protease enzyme active site (where the chopping takes place).
- The amino acids present at the enzyme’s active site.
- The electrostatic charge around the active site.
Additional factors had to be considered in order to design an effective treatment. For example, the compound’s solubility would determine its bioavailability when taken orally. Drugs that don’t absorb well in the body can increase the risk of drug resistance. Molecular modeling technology (which was new at the time) was used to modify the drug’s chemical makeup to design more effective treatment candidates.
One drug like AZT, it was good for a short while to suppress the virus. And then the virus figured out how to escape it. It was very clear we had to get combinations of different targets.”
— Anthony Fauci, National Institute of Allergy and Infectious Diseases, 2012, The Plague of Our Time: HIV/AIDS Epidemic, documentary by New England Journal of Medicine
During the late ’80s, there was a sense of extreme urgency to find solutions to the puzzles of HIV infection. The clock was clearly ticking: The HIV/AIDS death toll was increasing rapidly, not just in the U.S., but globally. Treatment activists clamored for changes in the FDA rules that governed clinical trials. They wanted more rapid testing of potential new treatments and broader access to investigational drugs for people already infected by HIV. Leading activists like Spencer Cox, Martin Delaney, Mark Harrington and many others were working with scientists to design and organize multi-drug trials.
One drug candidate proved to be potent and somewhat bioavailable as an oral drug. It failed in early testing, but Merck viewed it as an important milestone that confirmed they were on the right track. A later iteration combined one of Merck’s inhibitor candidates with one of Roche’s. Together, researchers then created an even better hybrid.
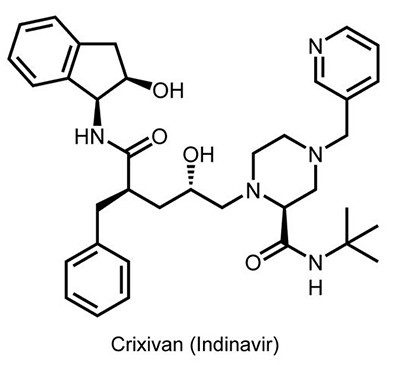
Pharmaceutical companies were inching closer to their goal. Merck refined their hybrid design and landed on a molecule that they called L–735,524 (Crixivan®, or indinavir). It was soluble and potent in combating HIV, even when taken orally.
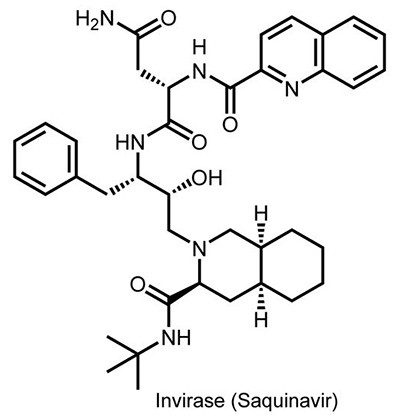
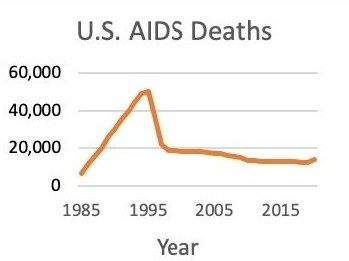
Roche moved their protease inhibitor, saquinavir (Invirase®), to clinical trials in 1992. Merck’s indinavir started trials in 1993. At the time, more than 40,000 people with AIDS were dying in the U.S. every year. Worldwide, that annual toll stood at more than 700,000.
Merck’s trial hit a speed bump the following year: HIV adapted to resist the drug. Yet, in their data, the researchers noticed that one patient had undetectable levels of HIV.
Once again, Merck did not give up on indinavir. The company decided to run a trial combining the protease inhibitor with two reverse transcriptase inhibitors, 3TC and AZT. (3TC, also known as Epivir® and lamivudine, was discovered by Bernard Belleau of McGill University and developed and marketed by BioChem Pharma. It was approved by the FDA for use in combination with AZT in November 1995.) This time, 90% of patients had no detectable HIV after receiving the treatment for several weeks. The results demonstrated the power of combining drugs that attack different parts of HIV’s replication process.
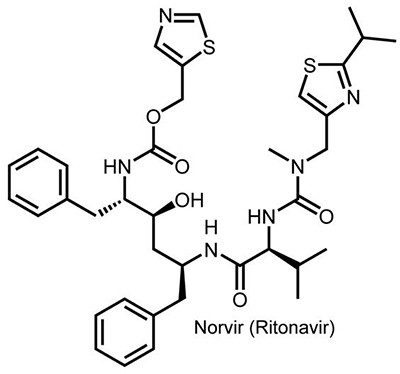
The FDA approved the first three protease inhibitors — saquinavir (Roche), indinavir (Merck), and ritonavir (Abbott’s Norvir®) — in late 1995 and early 1996. By 2000, Abbott (which spun off its pharmaceutical business as AbbVie in 2013) developed another protease inhibitor, lopinavir, and earned approval for use in children. And the company combined its two protease inhibitors in one drug, called Kaletra®. Today at least 10 protease inhibitors have been approved by the FDA.
HAART-felt
Doctors began prescribing protease inhibitors with reverse transcriptase inhibitors in 1996. The one-two punch was called highly active antiretroviral therapy, or HAART. It proved more effective than any one HIV drug on its own. HAART consistently lowers the amount of HIV present in the blood to undetectable levels within weeks.
Almost immediately after gaining regulatory approval for treating HIV, HAART began saving lives. A study from 1998 estimated that HAART had cut the U.S. AIDS death rate by 70% since the epidemic peaked there in 1995. Worldwide, the AIDS death rate didn’t peak until 2003-05. By 2021, 28.7 million people were receiving HIV antiretrovirals globally, out of an estimated 38.4 million people with HIV, according to UNAIDS.
It’s been more than 25 years since the first HAART earned approval. These treatments proved that, for HIV, two defenses are better than one. The first generation of HAART controlled HIV’s protease and reverse transcriptase — essentially tying both of the virus’ hands behind its back. Newer antiretrovirals combine reverse transcriptase inhibitors with drugs that defend against other elements of HIV, like integrase enzymes that slip HIV’s instructions into human DNA.
Newer generations of antiretrovirals also offered improvements in safety, tolerability, convenience, and efficacy. Some of the early regimens required people to take as many as 36 pills a day, often on complicated dosing schedules — at specific times, with strict dietary restrictions. Today, the most common first-line combination regimens are usually one pill, once a day. Together, all these approaches give care providers different tools to combat a patient’s HIV infection.
At the time that HAART was introduced in North America and Western Europe, people in lower- and middle-income countries had less access to treatment. There were many reasons for this — the price of the early regimens was unaffordable in developing countries, health systems in many countries lacked the infrastructure and trained health workers to manage national treatment programs at scale, and stigma and discrimination kept many people from seeking care and treatment. Sub-Saharan Africa, for example, remains home to the majority of HIV infections — about 860,000 new infections per year, and some 5.5 million untreated infections.
Over time, and thanks to a global movement to improve access to prevention, care, and treatment to address the risks of the HIV pandemic, HAART became more widely available and affordable. Pharmaceutical companies provided their products at tiered prices and licensed them to generic manufacturers, which were able to make lower-priced versions of the leading HAART medications available. In lower- and middle-income countries, the 2003 U.S. President’s Emergency Plan for AIDS Relief and the Global Fund to Fight AIDS, Tuberculosis and Malaria played central roles in this transformation, bringing new resources to the fight and helping create a functioning marketplace for antiretroviral treatment in Sub-Saharan Africa and other regions of the developing world.
HAART isn’t perfect — no drug regimen is. The treatments come with digestive and other side effects, and they’re not a cure. The virus can become resistant if a person doesn’t take their pills as prescribed. However, progress continues, and in 2021, FDA approved a treatment that works via bi-monthly injections. It combines reverse transcriptase inhibitors with an integrase inhibitor.
Today’s combination treatment regimens allow people with HIV who have access to treatment to live into their 70s — a huge win making HIV a manageable chronic disease. HIV’s antiretrovirals help avert over 1 million deaths every year. Yet this achievement didn’t happen overnight. The last 30 years were full of false starts and failures. But the story of HAART is an example where science, through the dedication and collaboration of skilled researchers, successfully addressed a global public health crisis.
Glossary
Enzyme: Typically identifiable by name because the word has the suffix -ase, an enzyme is a biological molecule that is responsible for speeding up chemical reactions. For example, reverse transcriptase is the enzyme in HIV that is responsible for converting an RNA template into DNA.
Highly active antiretroviral therapy (HAART): This combination treatment includes multiple drugs that fight HIV through different mechanisms. Also referred to as antiretroviral therapy (ART).
Integrase inhibitors: These antiretroviral drugs disable integrase, an enzyme that HIV uses to insert its genetic material into CD4+T cells. That stops HIV from replicating.
Protease inhibitors: These antiretroviral drugs block protease (an HIV enzyme). That prevents new (immature) HIV from becoming a mature virus that can infect other CD4+T cells.
Reverse transcriptase inhibitors: These antiretroviral drugs bind to and block HIV reverse transcriptase (an HIV enzyme). HIV uses reverse transcriptase to translate its RNA into DNA (reverse transcription). Blocking this process stops HIV from replicating.
I was in the intensive care unit, in a coma. The doctors had given me less than 24 hours to live. I went on HAART, and got better. And the rest is history in the making.”
— Phill Wilson, Black AIDS Institute, 2012, The Plague of Our Time documentary by New England Journal of Medicine
[M]illions of people have had their lives prolonged and saved by available therapy.”
— James Curran, Emory University, 2011, Remembering 30 Years of HIV/AIDS: Dr. James Curran, video from AIDS.gov
Landmark dedication and acknowledgments
Landmark dedication
The American Chemical Society (ACS) honored the discovery of highly active antiretroviral therapy for HIV with the National Historic Chemical Landmark (NHCL) designation in a ceremony at Merck in West Point, Pennsylvania, on Oct. 7, 2022, and at GSK in Collegeville, Pennsylvania, on May 15, 2024. The commemorative plaque reads:
The human immunodeficiency virus (HIV) was identified as the cause of acquired immunodeficiency syndrome (AIDS) in 1984, and the first treatment for the disease, AZT, was approved by the U.S. Food and Drug Administration in 1987. However, HIV quickly developed resistance to this drug, and deaths climbed. Pharmaceutical researchers at Abbott, Burroughs Wellcome (now part of GSK), Merck, Roche, and elsewhere sought new treatments to attack the virus through multiple mechanisms. Over the next decade, they developed drugs that work synergistically to defend people from the infection when used in combination. This highly active antiretroviral therapy (HAART) has saved millions of lives worldwide and, for those with access to treatment, transformed a once-seemingly undefeatable pandemic into a manageable chronic disease.
Acknowledgments
Written by Max Levy.
The author wishes to thank contributors to and reviewers of this booklet, all of whom helped improve its content, especially members of the ACS NHCL Subcommittee.
The nomination for this Landmark designation was prepared by Merck, with support from the ACS Division of Medicinal Chemistry, the ACS Philadelphia Section, GSK, AbbVie, and Roche.