Raney® Nickel: A Life-Changing Catalyst
Dedicated at the University of Tennessee at Chattanooga on April 7, 2022.
Murray Raney (1885-1966) invented the catalyst now known as Raney® nickel in 1926 while working out of his home laboratory near Chattanooga, Tennessee. Initially used to turn liquid vegetable oils into solid shortening, Raney nickel subsequently found applications in many industrial processes. By 1950 it had become one of the most widely utilized catalysts in the world, converting building-block chemicals into products that are an integral part of modern daily life, such as pharmaceuticals, food ingredients, agrochemicals, fibers, fragrances and personal care products. In 1963, W. R. Grace & Co. bought the company Raney had created, and to this day it continues to produce the catalyst at its manufacturing plant in Chattanooga.
Contents

Hydrogenation
Raney nickel is a heterogeneous hydrogenation catalyst. It promotes the reaction of hydrogen with organic compounds (compounds that contain carbon), and, like all catalysts, is not consumed in the process. Heterogeneous catalysts, like Raney nickel, are insoluble and easily recovered after reaction, so they can be used again and again.
Sporadic reports about hydrogenation reactions began cropping up in the 19th century, but the first full account of the addition of hydrogen to organic compounds did not appear until an 1897 publication by French scientist Paul Sabatier (1854-1941).
The first significant application of this hydrogenation technique was the conversion of liquid vegetable and seed oils into shortening, a semi-solid fat that is less likely than oils to go rancid and therefore has a longer shelf life. Shortening also proved to be a cheaper cooking alternative to rendered pork fat or lard, and its popularity soared. Worldwide, billions of pounds of liquid vegetable and seed oils are now hydrogenated into shortening and shortening-type products each year.
But that enormous expansion might not have happened without Raney, a University of Kentucky-educated engineer. In 1915, the Lookout Oil & Refining Co. of Chattanooga hired him to design, install and maintain electrolytic cells used to produce hydrogen for hydrogenating cottonseed oil. Raney became interested in the hydrogenation process, which employed a powdered nickel catalyst similar to that used by Sabatier. Raney’s fascination with the process led him to investigate the catalyst itself.
He experimented extensively in his home laboratory, initially on Oak Street in Chattanooga and then on nearby Lookout Mountain after he moved in 1922. There, in 1924, Raney developed a better nickel-based catalyst that hydrogenated cottonseed oil four times faster than the best powdered nickel catalyst. This new nickel catalyst was prepared by etching silicon from an alloy of nickel and silicon with sodium hydroxide. This “digestion” process produced holes in the alloy particles, similar to the holes in sponges. The result was a much larger surface area and greater number of active catalytic sites, boosting the catalyst’s reactivity.
Two years later, Raney created a new nickel catalyst, based on a similar process but using a nickel-aluminum alloy. This active nickel catalyst, patented in 1927, became known as Raney nickel.
Raney nickel offers some major advantages. For one, it’s considerably less expensive than the alternative: catalysts made with precious metals like platinum and palladium. Furthermore, its catalytic properties can be modified by varying the parameters of the digestion process. The activity and selectivity of Raney nickel can be further fine-tuned with the addition of small amounts of inexpensive metals such as chromium, iron, copper or molybdenum. These additives determine where hydrogen will be added to molecules with more than one potential insertion site.

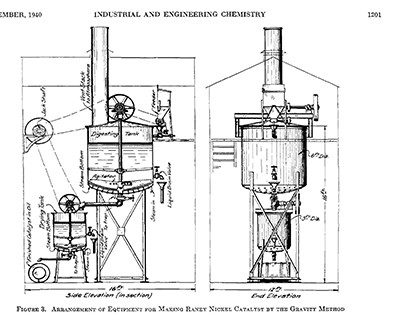
View larger image
Belying his future success, Raney, who didn’t bother going to high school, described himself at that age as “‘a wild boy without purpose in life,’ only a whisper away from a modern juvenile delinquent.” He later earned a degree at the University of Kentucky."
— Chattanooga Times, Sept. 2, 1963
Impact of Raney nickel
Although developed for the hydrogenation of vegetable oils, Raney nickel has become one of the most widely used hydrogenation catalysts in the world, yielding millions of tons of consumer and industrial products each year, including pharmaceuticals, sugar substitutes, solvents and personal care products.
Consumers are impacted by the catalyst’s discovery every day. For instance, when they use a toothbrush, walk on carpet or sit on fabric seats, they are probably making use of nylon bristles or fibers. A key intermediate in nylon production is cyclohexane, which is made from benzene via hydrogenation with Raney nickel. Benzene itself was initially derived from coal and is now obtained from petroleum.
In fact, Raney nickel contributed to the development of the petrochemical industry, which emerged in the U.S. after World War I (1914-18) and became a major economic factor after World War II (1939-45). During World War II, production of Raney nickel was considered a high priority for the U.S. because of the catalyst’s role in the fight against malaria, a serious and sometimes fatal disease picked up by military personnel (and others) in tropical regions. When supplies of quinine — the traditional malaria treatment — ran short, the catalyst was used in the preparation of the quinine substitute Atabrine.
Raney nickel’s ability to convert petroleum-based building-block chemicals into value-added materials is further illustrated with the following examples:
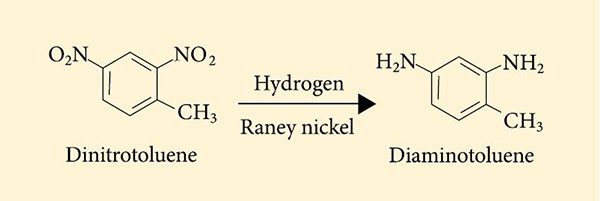
- Petroleum-derived toluene is nitrated to dinitrotoluene followed by hydrogenation with Raney nickel to make diaminotoluene. This compound is used to make polyurethane foam for seat cushions, mattresses, upholstered furniture, carpet backing and other products.
- Polyesters are a major class of polymers used extensively in fabric production. Butanediol is an important component in the preparation of polyesters, and it is made by hydrogenating butynediol with Raney nickel. Butynediol is made from acetylene, which in turn is generated from methane.
- Sulfolane is an industrial solvent used to refine petroleum and to remove unwanted gases during natural gas production. Sulfolane is made from sulfolene via hydrogenation with Raney nickel. Sulfolene, in turn, is made from butadiene obtained from petrochemical feedstocks.
An important non-petroleum example is sorbitol. This sugar substitute is produced by using Raney nickel to hydrogenate the sugar glucose, which is made from starch from sources such as potatoes or corn. Sorbitol is commonly used as a sweetener in toothpastes, mouthwashes, and sugar-free gums and candies because it does not support growth of the oral bacteria that cause cavities.
Raney nickel has numerous other applications in making products that consumers rely on every day, further demonstrating that the international recognition Raney nickel and its inventor Murray Raney have earned is richly deserved.


[Raney’s] discovery turned out to be a catalyst which has revolutionized chemical processes in a wide variety of industries ranging from petroleum to pharmaceutical, from the food industry to plastics, and in medical research.”
— Chattanooga Times, Dec. 25, 1964
Landmark dedication and acknowledgments
Landmark dedication
The American Chemical Society (ACS) honored the development of Raney® nickel with a National Historic Chemical Landmark (NHCL) designation in a ceremony at the University of Tennessee at Chattanooga on April 7, 2022. The commemorative plaque reads:
Working out of his home laboratory located near this site, Murray Raney (1885-1966) discovered the catalyst now known as Raney nickel in 1926. The discovery enabled the production of solid shortening from liquid vegetable oils. Raney nickel proved useful for this and many other industrial processes, becoming one of the most widely utilized catalysts in the world for conversion of building-block chemicals into pharmaceuticals, food ingredients, agrochemicals, fibers, fragrances and personal care products. Products made using Raney nickel are encountered daily. W. R. Grace & Co. bought Raney’s company in 1963 and continues to produce the catalyst at its manufacturing plant in Chattanooga, Tennessee.
Acknowledgments
The RANEY® brand of catalysts are trademarks, registered in the U.S. and/or other countries, of W. R. Grace & Co.
Written by Robert Mebane, emeritus, University of Tennessee at Chattanooga.
The author wishes to thank contributors to and reviewers of this booklet, all of whom helped improve its content, especially members of the ACS NHCL Subcommittee.
The nomination for this Landmark designation was prepared by the University of Tennessee at Chattanooga, the Chattanooga Section of the ACS, and W. R. Grace.

Booklet PDF
Additonal resources
Further reading
- “Murray Raney of Chattanooga and Nickel Catalysts” (ACS)
- “The Discoverer of Raney Nickel” (ACS)
- “S. R. Murray Raney – Pioneer Catalyst Producer” (ACS)
- “History of Petroleum Chemicals Industry” (ACS)
- “UTC to commemorate one of only three ‘landmark’ chemical discoveries in Tennessee” (University of Tennessee at Chattanooga)
- “Wallace Carothers and the Development of Nylon” (ACS)

