What molecule am I?
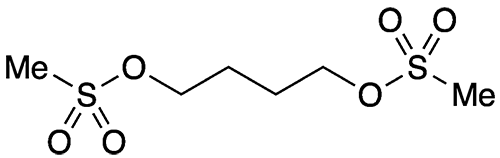
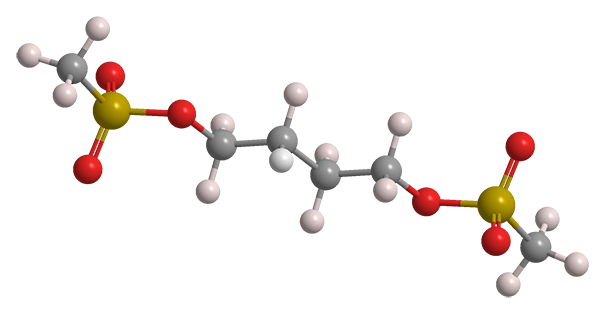
Busulfan, a common name for the dimethanesulfonate ester of 1,4-butanediol, is an older chemotherapy drug sold under the trade names Myleran, Busulfex, and many others. It was described as a leukemia treatment by inventor Geoffrey M. Timmins in a 1959 US Patent to Burroughs Wellcome (Tuckahoe, NY; now part of GlaxoSmithKline).
Busulfan’s mode of action is as a bifunctional alkylating agent, which has an immunosuppressive effect on bone marrow. It stops tumor growth by cross-linking guanine bases in DNA double-helix strands. This makes the strands unable to uncoil and separate. Because this process is necessary for DNA replication, the tumor cells can no longer divide. In addition, busulfan adds methyl groups where they do not belong, which leads to DNA miscoding.
Almost a decade before the Timmins patent was issued, busulfan was implicated in the death of a 67-year-old breast cancer patent. An anonymous 1950 article in the British Medical Journal titled “Death after new drug” compared busulfan to the properties of nitrogen mustards, war gases that were also being tried as anticancer agents. The article suggested that busulfan behaved anomalously in the body compared with other compounds with similar properties.
Despite this mishap and its dangerous properties (see the hazard information table), busulfan continued to be developed as a cancer drug. In 1999, it was approved by the US Food and Drug Administration for treating chronic myeloid leukemia. Its use was soon reduced because of the emergence of Novartis’s imatinib1, which was approved by the FDA for treating cancer 2 years after busulfan. Imitanib, however, has its own adverse effects and is more expensive, and busulfan is still on the market. It is currently listed as an orphan drug by the FDA.
1. CAS Reg. No. 152459-95-5.
Busulfan hazard information*
| Hazard class** | GHS code and hazard statement | |
|---|---|---|
| Acute toxicity, oral, category 3 | H301—Toxic if swallowed | |
| Acute toxicity, dermal, category 2 | H310—Fatal in contact with skin | |
Acute toxicity, inhalation, category 2 | H330—Fatal if inhaled | |
| Germ cell mutagenicity, category 1B | H340—May cause genetic defects | |
| Carcinogenicity, category 1A | H350—May cause cancer | |
*Compilation of multiple safety data sheets. The hazard statements given here are the most severe reported.
**Globally Harmonized System (GHS) of Classification and Labeling of Chemicals. Explanation of pictograms.
Molecule of the future
In 2021, Scott E. Collibee and a team of 24 researchers at Cytokinetics (South San Francisco, CA) reported the discovery of reldesemtiv1, which they described as “a second-generation fast skeletal muscle troponin activator . . . that increases force production at submaximal stimulation frequencies.” It is targeted at treating impaired muscle function in patients with amyotrophic lateral sclerosis, or Lou Gehrig’s disease.
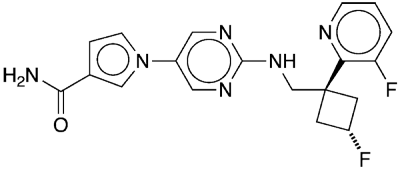
At the time the researchers’ article was published, reldesemtiv was in a phase 1 (safety screening) clinical trial. By August 2021, the prospective drug had already moved to a phase 3 trial to evaluate its efficacy and safety in a group of 555 human subjects.
1. CAS Reg. No. 1345410-31-2.
Molecule of the Future
Once a month we bring you a newly discovered or developed molecule that has important implications for the future of chemistry or society in general. Look for it the third week of each month. Learn more about this month's Molecule of the Future below.
We're looking for more molecules of the future!
Do you have a suggestion for the next molecule of the future? Send your idea to MOTW.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Busulfan fast facts
| CAS Reg. No. | 55-98-1 |
| SciFinder nomenclature | 1,4-Butanediol, 1,4-dimethanesulfonate |
| Empirical formula | C6H14O6S2 |
| Molar mass | 264.30 g/mol |
| Appearance | White to beige crystals or powder |
| Melting point | 114–118 °C |
| Water solubility | ≈9 mg/L (dec.) |
MOTW update
Pentachlorophenol was the Molecule of the Week for July 19, 2021. It is an old-line insecticide and wood preservative that is a “reasonably anticipated” human carcinogen and an environmental hazard.
After decades of restricting the use of pentachlorophenol, the US Environmental Protection Agency proposed a complete ban on it in early 2021. This month, EPA followed through on the ban, which will go into effect in 2024. Wood-treatment companies are permitted to use up existing stocks until 2027.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

