What molecule am I?

March is MOTW Solvent Month! This is the second of four articles about key solvents—Ed.
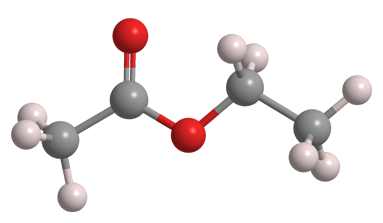
Ethyl acetate is one of the simplest carboxylate esters. (Former Molecule of the Week methyl formate is the simplest.) The colorless liquid has a sweet, fruity odor that most people find pleasant.
As you might expect, ethyl acetate was first synthesized from ethanol and acetic acid. The reaction was the classic acid-catalyzed Fischer esterification, which dates back to 1895. This is still the most widely used commercial synthesis. An alternative method is the Tishchenko reaction in which acetaldehyde disproportionates in the presence of base to the alcohol and the acid that then esterify in situ.
Ethyl acetate is a widely used solvent, especially for paints, varnishes, lacquers, cleaning mixtures, and perfumes. Like last week’s MOTW, dichloromethane, it is used as a solvent for decaffeinating coffee beans. In the lab, ethyl acetate is a common solvent for column and thin-layer chromatography.
Ethyl acetate hazard information
| GHS classification*: Flammable liquids, category 2 | |
| H225—Highly flammable liquid and vapor | |
| GHS classification: serious eye irritation, category 2A | |
| H319—Causes serious eye irritation | |
| GHS classification: specific target organ toxicity, single exposure, central nervous system, category 3 | |
| H336—May cause drowsiness or dizziness | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Ethyl acetate fast facts
| CAS Reg. No. | 141-78-6 |
| Empirical formula | C4H8O2 |
| Molar mass | 88.11 g/mol |
| Appearance | Colorless liquid |
| Boiling point | 77 ºC |
| Water solubility | 83 g/L |
MOTW update
N,N-Dimethyltryptamine (DMT) was the Molecule of the Week for September 17, 2018. DMT is a plant-based hallucinogen that is outlawed in most countries; but researchers are exploring its possible use as an antidepression drug. Now, David E. Olson at the University of California, Davis, and colleagues at several institutions report that “chronic, intermittent, low doses of DMT produced an antidepressant-like phenotype and enhanced fear extinction learning without impacting working memory or social interaction” in lab rats. Unfortunately, the rats also gained significant amounts of body weight. Their research continues.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.


