What molecule am I?
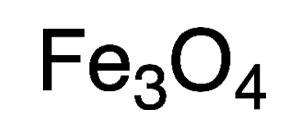
Magnetite is a mineral whose primary component is an iron oxide that contains equal amounts of iron(II) and iron(III). Its empirical formula is Fe3O4, and it is often expressed as iron(II,III) oxide. In the past, it has been called ferrous–ferric oxide and triiron tetraoxide.
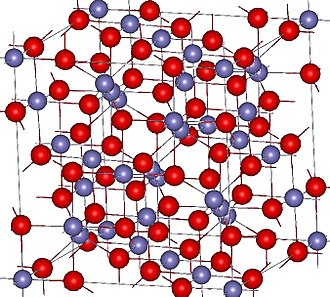
Magnetite is found in igneous, metamorphic, and sedimentary rocks. As its name implies, it is magnetic; it and other inherently magnetic iron-containing minerals are described as being ferrimagnetic. Magnetite is in the hexoctahedral crystal class; in the 3-D crystal structure, the purple atoms are iron, and the red atoms are oxygen.
Magnetite’s greatest use is as an important iron ore for steel manufacture. Other applications are as a catalyst in the Haber process for making ammonia, as a pigment for paints and ceramics, and as magnetic micro- and nanoparticles for a variety of processes and materials.
Studying an intriguing application of magnetite in nature, Lewis C. Naisbett-Jones, David L. G. Noakes, and colleagues at the University of North Carolina (Chapel Hill), LGL Ecological Research Associates (Bryan, TX), Oregon State University (Corvallis), and the Oregon Hatchery Research Center (Alsea) discovered that salmon use tiny magnetite crystals in their tissues to help them make their way from far out in the ocean back to their inland spawning locations.
The researchers subjected juvenile chinook salmon to strong magnetic pulses to show that the fish oriented their movements according to the direction of the magnetic field. Their results led the researchers to conclude that salmon are guided by Earth’s magnetic field to return to the rivers where they spawned.
Magnetite hazard information*
| Hazard class** | Hazard statement |
|---|---|
| Not a hazardous substance or mixture |
*Information for powder.
**Globally Harmonized System of Classification and Labeling of Chemicals.
Magnetite fast facts
| CAS Reg. No. | 1317-61-9 |
| SciFinder nomenclature | Iron oxide (Fe3O4) |
| Empirical formula | Fe3O4 |
| Molar mass | 231.53 g/mol |
| Appearance | Gray to black crystals or black powder |
| Melting point | 1583–1597 ºC |
| Water solubility | Insoluble |
MOTW Update
Carbonyl sulfide (COS or OCS) was the Molecule of the Week for July 24, 2017. It is a colorless gas with a structure similar to that of carbon dioxide (CO2). Although there is only a tiny amount of COS in the atmosphere, researchers found that it is a good surrogate for CO2 for tracking how much carbon is taken up by photosynthesis worldwide.
In an upcoming book titled “Slaying the Climate Dragon: Energy Systems as Our Sword & Shield”, Sandy (George) Lawrence writes, “Carbonyl sulfide is co-absorbed with CO2 into plants, where the cells rapidly deactivate the OCS but utilize the CO2. The amount of OCS thus represents an inverse marker of aggregate global photosynthetic activity. The record in glacial ice bubbles of miniscule amounts represents a record that in theory goes back 600,000 years in Greenland ice cores [and more than] 800,000 years in Antarctic ice cores.”

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

