What molecule am I?
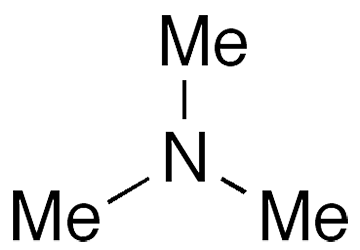
Trimethylamine (TMA) is the simplest tertiary aliphatic amine. It’s responsible for the pungent odor given off by fish and other marine and aquatic animals. It is also a degradation product of plants that have high nitrogen contents and a distillation product of sugarbeet residues.
Trimethylamine has been known since at least 1878, but it is uncertain as to who discovered and identified it. Several 19th-century patents and journal articles describe its use for converting chloride salts to carbonates and bicarbonates, as a reagent in the dye industry, and in other chemical reactions.
Trimethylamine is a gas at ambient temperature, but it is usually sold as a liquid under pressure or in an aqueous solution. It can be dangerous to handle, as shown in the hazard information table. It is a strong Lewis base that forms adducts with Lewis acids such as borane and other hydrides, metal halides, and related organometallic compounds.
Trimethylamine is used as a basic catalyst, in the preparation of quaternary ammonium hydroxides and salts, and in ion-exchange resins. It is also an important starting material for manufacturing the essential nutrient choline1; conversely, excessive choline intake results in its degradation to TMA in the body.
The 2022 worldwide market value of trimethylamine is estimated at US$109 million. At ≈$2250/t, this year’s production should approach 50,000 tonnes.
1. CAS Reg. No. 62-49-7.
Trimethylamine hazard information
| Hazard class* | GHS code and hazard statement | |
|---|---|---|
| Flammable gases, category 1 | H220—Extremely flammable gas | |
| Gases under pressure, liquefied gas | H280—Contains gas under pressure; may explode if heated | |
| Skin corrosion/irritation, category 2 | H315—Causes skin irritation | |
| Serious eye damage/eye irritation, category 1 | H318—Causes serious eye damage | |
| Acute toxicity, inhalation, category 4 | H332—Harmful if inhaled | |
| Specific target organ toxicity, single exposure, respiratory tract irritation, category 3 | H335—May cause respiratory irritation | |
*Globally Harmonized System (GHS) of Classification and Labeling of Chemicals. Explanation of pictograms.
Molecules from the journals
Two substances that may have therapeutic effects were discovered in 2009 in water taken from an abandoned South Korean coal mine. (–)-Glionitrin A1 has antibacterial and antitumor activity; the closely related (–)-glionitrin B2 can hamper the migration of cancer cells. The total syntheses of both were reported in 2021 by Nicolas R. Koning, Anders P. Sundin, and Daniel Strand* at Lund University (Sweden).
Rongalite3, or sodium hydroxymethanesulfinate, a salt known since 1908, has long been used as a bleaching and reducing agent in the dye industry. This year, Yan-Dong Wu, An-Xin Wu, and colleagues at Central China Normal University (Wuhan) showed that it can function as a sulfone source, C1 synthon, and radical initiator—all in the same reaction.
1. CAS Reg. No. 1116153-15-1.
2. CAS Reg. No. 1155759-94-6.
3. CAS Reg. No. 149-44-0.
New Feature!
Molecules from the journals
MOTW is happy to launch Molecules from the journals, in which we briefly describe noteworthy molecules that appeared in recent ACS journal articles. See the first edition below.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Trimethylamine
fast facts
| CAS Reg. No. | 75-50-3 |
| SciFinder nomenclature | Methanamine, N,N-dimethyl- |
| Empirical formula | C3H9N |
| Molar mass | 59.11 g/mol |
| Appearance | Colorless gas or liquid |
| Boiling point | 3 °C |
| Water solubility | Miscible |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

