What molecule am I?
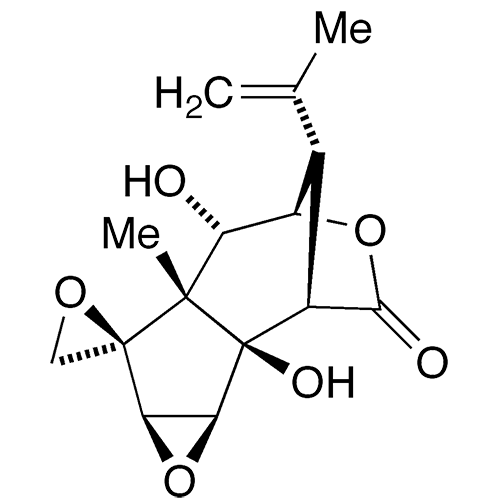
Tutin is a tetracyclic organic molecule with nine chiral centers. It exists in nature in six species of the tutu plant (genus Coriara) that grows in New Zealand. It is extremely toxic.
Tutin and its fellow toxin coriamyrtin1 were first described in 1901 by Thomas Hill Easterfield at Victoria College and Bernard Cracroft Aston at the New Zealand Department of Agriculture (both in Wellington). These compounds, along with a third tutu ingredient, picrotoxinin2, were responsible for poisoned honey that began to afflict New Zealanders in the 19th century.
Honeybees (Apis mellifera) don’t get it from the tutu plant’s pollen or nectar; the toxins are present only in the green plant parts. So how do tutin and its cousins get into honey? According to New Zealand’s Ministry for Primary Industries, “Passion vine hopper insects [Scolypopa australis] feed on tutu plants and produce honeydew which contains tutin. When bees collect this honeydew, the honey they make can contain tutin.”
Tutin’s toxicity results from its ability to inhibit glycine receptors in spinal neurons. Glycine is an inhibitory neurotransmitter; when it is inhibited, individuals can exhibit hypersalivation, agitation, seizures, exhaustion, coma, and respiratory distress. These signs are often preceded by nausea, blurred vision, and memory deficit. As of 2017, no specific antidote had been developed; recommended treatment is diazepam or barbiturates to control seizures and maintenance of respiratory function.
In 2016, the Ministry for Primary Industries issued an extensive guide for beekeepers, packers, and exporters to comply with New Zealand’s food standard for tutin in honey.
1. CAS Reg. No. 2571-86-0.
2. CAS Reg. No. 17617-45-7.
Tutin hazard information*
| Hazard class** | GHS code and hazard statement | |
|---|---|---|
| Acute toxicity, oral, category 3 | H301—Toxic if swallowed | |
*Hazard information difficult to obtain; this information may be incomplete.
**Globally Harmonized System (GHS) of Classification and Labeling of Chemicals. Explanation of pictograms.
Molecules from the journals
N1-(1,3-Dimethylbutyl)-N4-phenyl-1,4-benzenediamine1—commonly known as 6DDP—is a commonly used antioxidant and antiozonant additive in rubber tires. Its quinone ozonation product, 6PPDQ2, has been shown to contribute to the die-off of coho salmon (Oncorhynchus kisutch) and other commercial fish as the result of stormwater runoff from roads into streams. Last year, Erik T. Krogh and colleagues at institutions in British Columbia and California reported a direct mass-spectrometry method for the rapid analysis of 6PPDQ in environmental samples.
Quassin3 is one of a series of bitter-tasting terpenoids that are extracted from the wood of the quassia tree (Quassia amara) native to Central and South America. Even though it is one of the bitterest substances known, quassin is used to flavor foods and, most notably, Angostura bitters. In 2021, William P. Thomas and Sergey V. Pronin* at the University of California, Irvine, reported a concise, enantioselective synthesis of quassin and other quassinoids.
1. CAS Reg. No. 793-24-8.
2. CAS Reg. No. 2754428-18-5.
3. CAS Reg. No. 76-78-8.
Molecules from the Journals
MOTW briefly describes noteworthy molecules that appeared in recent ACS journal articles. See this week's
edition below.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Tutin fast facts
| CAS Reg. No. | 2571-22-4 |
| SciFinder nomenclature | Spiro[2,5-methano- 7H-oxireno[3,4]cyclopent[1,2-d]oxepin-7,2′-oxiran]-3(2H)-one, hexahydro-1b,6-dihydroxy-6a-methyl-8-(1-methylethenyl)-, (1aS,1bR,2S,2′R,5R,6S,6aR,7aR,8R)- |
| Empirical formula | C15H18O6 |
| Molar mass | 294.30 g/mol |
| Appearance | Colorless crystals |
| Melting point | 212–213 °C |
| Water solubility | Soluble; specific solubility not reported |
MOTW update:
November 27, 2023
6PPD1—full name N1-(1,3-dimethylbutyl)-N4-phenyl-1,4-benzenediamine—was a Molecule from the Journals for May 2, 2022. An antioxidant and antiozonant additive in rubber tires, its ozonation product 6PPDQ2 contributes to the die-off of coho salmon and other commercial fish as the result of stormwater runoff from roads into streams. This month, the US Environmental Protection Agency agreed to hear a petition from three West Coast Indigenous people’s tribes that called for the agency to regulate the use of 6PPD in tires. Coho salmon are central to the economies and diets of the tribes and other nearby communities.
1. CAS Reg. No. 793-24-8.
2. CAS Reg. No. 2754428-18-5.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

