What molecule am I?

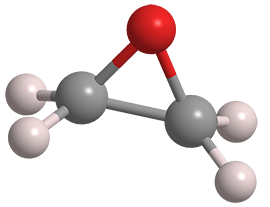
Ethylene oxide is the smallest of the oxirane family of molecules. It is a sweet-smelling, colorless gas that has many uses; but its high reactivity presents many hazards.
Back in 1859, A. Wurtz treated ethylene chlorohydrin with potassium hydroxide to make ethylene oxide and potassium chloride. In 1914, BASF built the first ethylene oxide plant, which used the chlorohydrin method, but with calcium hydroxide instead of KOH. This process was eventually superseded by the direct oxidation of ethylene, which is used exclusively today.
The uses of ethylene oxide are numerous. By far, its primary use is as a raw material for the industrial manufacture of ethylene glycol and its oligomers, glycol ethers, and ethanolamines. Minor, but important, direct applications include a fumigant for foods and textiles; an agricultural fungicide and insecticide; and a sterilant for medical equipment.
As shown in the hazard information table, many of ethylene oxide’s uses also make it extremely dangerous. Its carcinogenicity is of special concern. The US Environmental Protection Agency is under a court order to decide by March 13, 2020, whether to tighten the 2006 standards set for air pollution by ethylene oxide and other organic chemicals.
There is broad disagreement between environmental and industrial advocates as to the safe concentration of ethylene oxide in air. Proposals range from 0.1 ppt to 4 ppb—a factor of 40,000. Both sides are beginning to pressure EPA to see things their way.
Ethylene oxide hazard information
| GHS classification**: flammable gases, category 1 | |
| H220—Extremely flammable gas | |
| GHS classification: gases under pressure, liquefied gas | |
| H280—Contains gas under pressure; may explode if heated | |
| GHS classification: skin corrosion/irritation, category 2 | |
| H315—Causes skin irritation | |
| GHS classification: eye damage/eye irritation, category 2A | |
| H319—Causes serious eye irritation | |
| GHS classification: acute toxicity, inhalation, category 3 | |
| H331—Toxic if inhaled | |
| GHS classification: specific target organ toxicity, single exposure, respiratory tract irritation, category 3 | |
| H335—May cause respiratory irritation | |
| GHS classification: germ cell mutagenicity, category 1B | |
| H340—May cause genetic defects | |
| GHS classification: carcinogenicity, category 1B | |
| H350—May cause cancer | |
| GHS classification: hazardous to the aquatic environment, acute hazard, category 3 | |
| H402—Harmful to aquatic life | |
| GHS classification: hazardous to the aquatic environment, long-term hazard, category 3 | |
| H412—Harmful to aquatic life with long-lasting effects | |
*Data from Sigma–Aldrich; other vendors’ data vary.
**Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Ethylene oxide fast facts
| CAS Reg. No. | 75-21-8 |
| Empirical formula | C2H4O |
| Molar mass | 44.05 g/mol |
| Appearance | Colorless gas |
| Boiling point | 10.4–11.0 ºC |
| Water solubility | Miscible |
MOTW update:
May 04, 2020
Ethylene oxide is used extensively in chemical manufacturing, agriculture, and fumigation, but it is extremely hazardous. In March 2020, the US Environmental Protection Agency’s Office of the Inspector General found that the agency had not done enough to inform people who are living near ethylene oxide–emitting chemical plants that they are likely to have an increased risk of cancer. The OIG even recommended that EPA use unverified data to warn neighbors of the hazard.
MOTW update
Former Molecules of the Week 1,4-dioxane, hexabromocyclododecane, 1-bromopropane, N-methyl-2-pyrrolidone, and dichloromethane, among several other chemicals, are at the center of a controversy about how the EPA’s scientific advisory committees are evaluating risks of air pollution. The problems are the lack of expertise in some of the committees and a shortage of adquate data to evaluate the chemicals. Industrial and environmental stakeholders are critical of the latest risk assessments.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

