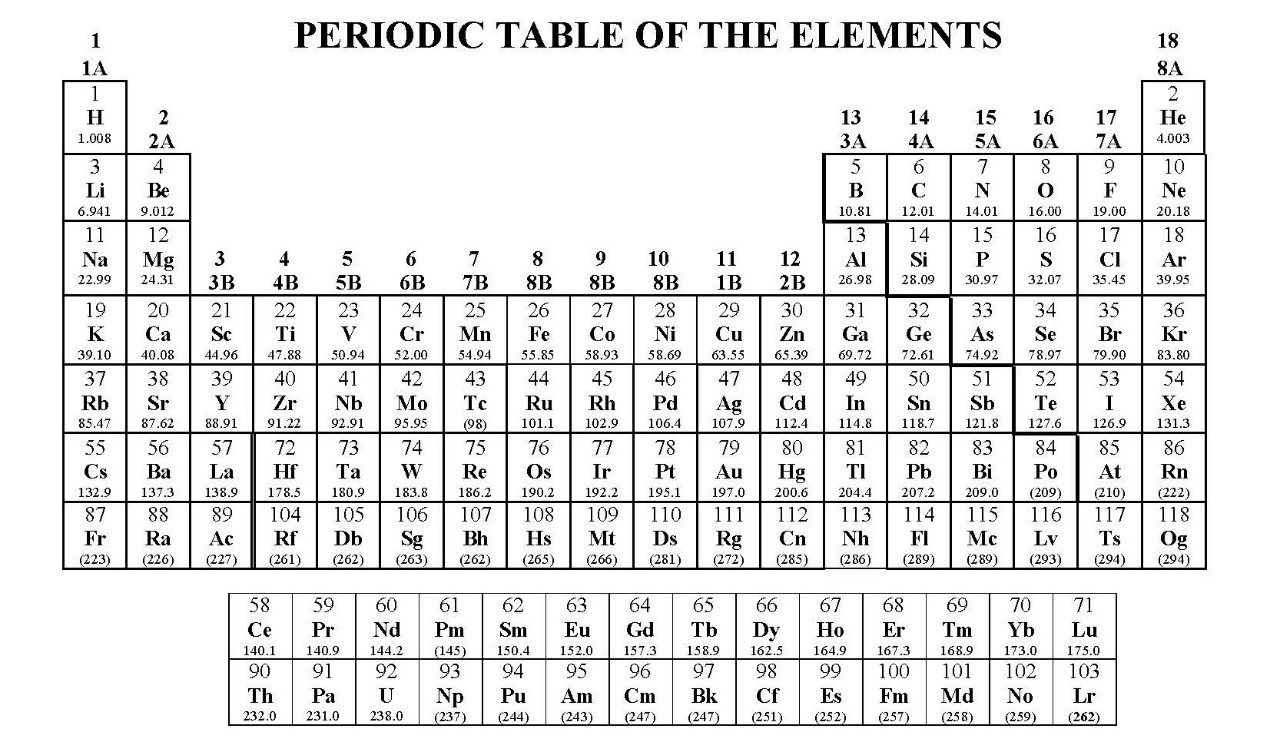
USNCO Exams Reference Page
Below is the standard information provided for reference in all USNCO exams. No additional information may be accessed while taking any USNCO exam.
Abbreviations and Symbols
| Full Name | Abbreviation/ Symbol |
|---|---|
| Amount Of Substance | N |
| Ampere | A |
| Atmosphere | Atm |
| Atomic Mass Unit | U |
| Avogadro Constant | NA |
| Celsius Temperature | °C |
| Centi– Prefix | C |
| Coulomb | C |
| Density | D |
| Electromotive Force | E |
| Energy Of Activation | Ea |
| Enthalpy | H |
| Entropy | S |
| Equilibrium Constant | K |
| Faraday Constant | F |
| Free Energy | G |
| Frequency | Ν |
| Gas Constant | R |
| Gram | G |
| Hour | H |
| Joule | J |
| Kelvin | K |
| Kilo– Prefix | K |
| Liter | L |
| Measure Of Pressure | Mm Hg |
| Milli– Prefix | M |
| Molal | M |
| Molar | M |
| Molar Mass | M |
| Mole | Mol |
| Planck’s Constant | H |
| Pressure | P |
| Rate Constant | K |
| Reaction Quotient | Q |
| Second | S |
| Speed Of Light | C |
| Temperature, K | T |
| Time | T |
| Vapor Pressure | VP |
| Volt | V |
| Volume | V |
| Year | Y |
Constants
R = 8.314 J mol–1 K–1
R = 0.08314 L bar mol–1 K–1
F = 96,500 C mol–1
F = 96,500 J V–1 mol–1
NA = 6.022 x 1023 mol–1
h = 6.626 x 10–34 J s
c = 2.998 x 108 m s–1
0 °C = 273.15 K
1 atm = 1.013 bar = 760 mm Hg
Specific heat capacity of H2O = 4.184 J g–1 K–1
Equations
E = Eo — (RT/nF) x lnQ
lnK = (—ΔHo/R) x (1/T) + constant
ln(k2/k1) = (Ea/R) x ((1/T1) — (1/T2))
Thank you to the Olympiad Examinations Task Force and the Olympiad Laboratory Practical Task Force. View all acknowledgements.