What molecule am I?
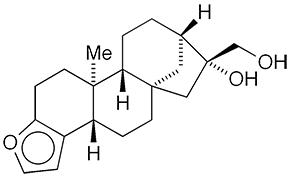

Cafestol1 is a diterpenoid found in coffee beans (Coffea arabica and other Coffea species). It is the reduced form of kahweol2, another coffee diterpenoid. Both diterpenoids exist partially as their esters in coffee beans.
In 1938, Karl Heinrich Slotta and Klaus Neisser at Instituto Butantan (São Paulo, Brazil) reported the isolation and identification of cafestol, which they found constituted 0.3–0.4 wt% of raw or roasted coffee. The authors weren’t able to obtain the air-sensitive kahweol, which had been reported 6 years earlier; but they did isolate γ-sitosterol3, a phytosterol that was identified in corn oil in 1926 by R. J. Anderson and R. L. Shriner at the NY Agricultural Experiment Station (Geneva).
Almost a half century after Slotta and Neisser’s work, Bruce C. Pettitt at the US Customs Service (Washington, DC), in a project to determine the geographical origin of coffee beans, examined the differences between the distributions of cafestol and kahweol esters in C. arabica and C. canephora, a species that grows in sub-Saharan Africa. Pettitt’s main finding was that the distribution is similar in both species. He also established that whereas kahweol is esterified with several acids, the only ester of cafestol is the palmitate.
Cafestol has been suspected of raising serum cholesterol and triglycerides in individuals who drink coffee, especially unfiltered coffee. In 2001, Martijn B Katan and coauthors at Wageningen University (The Netherlands), Glasgow Royal Infirmary (UK), and University Hospital Nijmegen (The Netherlands) reported on whether cafestol increases plasma triacylglycerol (triglycerides). In their eight-man study, they found that the subjects had increased plasma triacylglycerol by an average of 31%. They attributed the increase to a higher production rate of very-low-density-lipoprotein receptor-1 apolipoprotein B.
It was not until 2024 that a comprehensive study of the bioaccessibility of cafestol in boiled (unfiltered) coffee was done. Claudia Rezende and colleagues at the Federal University of Rio de Janeiro and other institutions in that city used an in vitro digestion model and liquid chromatography–high-resolution mass spectrometry to determine the fate of cafestol in the body. Their main results were that
- the bioaccessibility of cafestol in the intestinal phase of digestion was 93.65%;
- all cafestol is absorbed in its free alcohol form; its esters are enzymatically hydrolyzed; and
- a cafestol metabolite, 17-oxo-cafestol, was formed by oxidation during the oral phase of digestion.
1. SciFinder name: 5a,8-methano-5aH-cyclohepta[5,6]naphtho[2,1-b]furan-7-methanol, 3b,4,5,6,7,8,9,10,10a,10b,11,12-dodecahydro-7-hydroxy-10b-methyl-, (3bS,5aS,7R,8R,10aR,10bS)-.
2. CAS Reg. No. 6894-43-5.
3. CAS Reg. No. 83-47-6.
Cafestol hazard information
| Hazard class | GHS code and hazard statement |
|---|---|
| Not a hazardous substance or mixture |
Molecules from the Journals
Methanetriol1 [CH(OH)3], aka orthoformic acid, has been called an “impossible” molecule because the carbon atom bonded to three hydroxyl groups would spontaneously decompose to formic acid. But in 2024, Tao Yang at East China Normal University (Shanghai) and Shanxi University (China), Alexander M. Mebel at Florida International University (Miami), Ralf I. Kaiser at the University of Hawaii at Manoa (Honolulu), and collaborators at these and other institutions reported the synthesis of methanetriol.
The authors made the molecule by irradiating mixed methanol–molecular oxygen ices with energetic electrons at very low temperature (5 K). The irradiation produced methanetriol, along with its isomers MeO3H and CH2(OH)OOH. They confirmed the products after sublimation to the gas phase by using isomer-selective photoionization reflectron time-of-flight mass spectrometry.
L-Kynurenine2 is a natural amino acid. It is a metabolite of L-tryptophan3 that is an intermediate in the biosynthesis of nicotinic acid4 (niacin).
Last month, Yuhang Sui, Aimee J. Horsfall*, and Paul W. R. Harris* at the University of Auckland (New Zealand) reviewed the use of L-kynurenine to synthesize biologically active peptides. The authors cited the US FDA-approved antibiotic daptomycin5 as the most-studied example and discussed more recent discoveries, such as the taromycins (A6 and B7), as successful incorporations of the L-kynurenine structure in drugs.
1. CAS Reg. No. 463-78-5.
2. CAS Reg. No. 2922-83-0.
3. CAS Reg. No. 73-22-3.
4. CAS Reg. No. 59-67-6.
5. CAS Reg. No. 103060-53-3.
6. CAS Reg. No. 1567860-39-2.
7. CAS Reg. No. 2376885-37-7.
Molecules from the Journals
MOTW briefly describes noteworthy molecules that appeared in recent ACS journal articles. See this week's edition.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule that you would like us to consider, please send us a message, preferably including links to references about your suggested molecule. And thank you for your interest in Molecule of the Week! —Ed.
Cafestol fast facts
| CAS Reg. No. | 469-83-0 |
| Empirical formula | C20H28O3 |
| Molar mass | 316.44 g/mol |
| Appearance | White crystals or powder |
| Melting point | 160–162 °C (dec.) |
| Water solubility | Insoluble |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.


