What molecule am I?
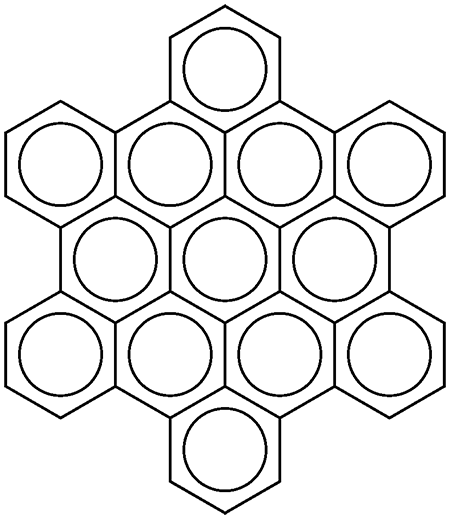
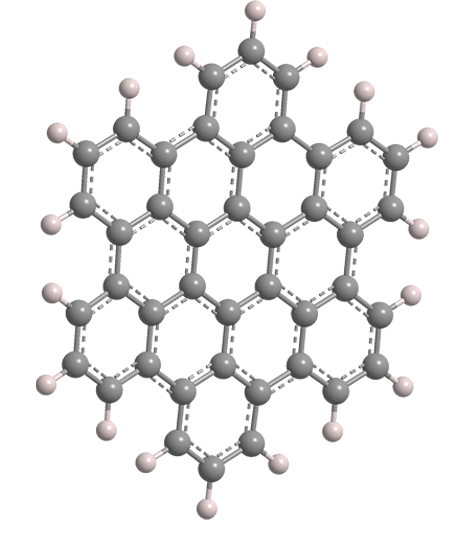
Hexa-peri-benzocoronene (HBC), or, more commonly, hexabenzocoronene, is a planar polycyclic aromatic hydrocarbon (PAH). Structurally, it consists of a central benzene ring surrounded first by six more benzene rings to form coronene1, which in turn is surrounded by six additional benzenes for a total of 13 rings. HBC is in the D6h symmetry group.
Two HBC syntheses appeared in 1958. In one, A. Halleux, R. H. Martin, and G. S. D. King at European Research Associates (Brussels) used two reactions to make the molecule: the cyclodehydrogenation of hexaphenylbenzene2 and the reaction of dibenz-1,9;2,3-anthrone3 with Zn/ZnCl2. They also isolated two new hydrocarbons from the Zn/ZnCl2 reaction.
In the other synthesis, E. Clar and C. T. Ironside at the University of Glasgow first treated 2,3,7,8-dibenzoperinaphthene with bromine, then heated the brominated product to form tetrabenzoperopyrene4 (TBP) Additional heating dehydrogenated TBP to produce HBC. The authors also reported that HBC emits orange-yellow phosphorescence in solid solution at low temperatures.
In 2014, Andreas Hirsch and collaborators at the University of Erlangen–Nuremberg (Germany) synthesized a compound in which HBC is covalently linked to buckminsterfullerene5. The authors stated that this “dyad” could serve as a versatile template for optoelectronic applications. X-ray crystallography showed that the bond distance between the two aromatic components is only 3.17 Å.
In another example of HBC and its derivatives combined with other chemical structures, Aleksey E. Kuznetsov at Federico Santa María Technical University (Santiago, Chile) paired HBCs with antiaromatic sulfur- and selenium-core-modified porphyrins (isophlorins). In this theoretical study, the author found that isophlorins had profound effects on the structural and electronic properties of the dyads and therefore likely their reactivities.
Although PAHs similar to HBC are known carcinogens, there are no reports in the literature of its carcinogenicity.
1. CAS Reg. No. 191-07-1.
2. CAS Reg. No. 992-04-1.
3. CAS Reg. No. 86854-05-9.
4. CAS Reg. No. 190-22-7.
5. CAS Reg. No. 99685-96-8; MOTW for June 11, 2018.
Hexa-peri-benzocoronene hazard information
| Hazard class* | GHS code and hazard statement | |
|---|---|---|
| Acute toxicity, oral, category 4 | H302—Harmful if swallowed | |
| Acute toxicity, dermal, category 4 | H312—Harmful in contact with skin | |
| Acute toxicity, inhalation, category 4 | H332—Harmful if inhaled | |
*Globally Harmonized System (GHS) of Classification and Labeling of Chemicals. Explanation of pictograms.
MOTW updates
Uranium hexafluoride1 (UF6) was the Molecule of the Week for February 28, 2011. It is a covalent compound used primarily for separating uranium isotopes. In a recent article, Kirk Peterson, Puru Jena, Kit H. Bowen, and colleagues at Johns Hopkins University (Baltimore), Washington State University (Pullman), and Virginia Commonwealth University (Richmond) used photoelectron spectroscopy and quantum chemistry to demonstrate the degree to which gold can act as a surrogate to fluorine in UF6. They found that, unlike UF6, UAu6 shows strong ligand–ligand (i.e., Au–Au) interactions that result in three low-lying isomers with 3-D or quasi-2-D structures.
Sulfuryl fluoride2 (SO2F2) was the Molecule of the Week for March 23, 2009. It is a relatively unreactive gas that is commonly used as a fumigant for controlling termites. It is also a greenhouse gas 4800 times more potent than CO2 with high atmospheric persistence. In October, two environmental organizations filed a legal petition with the California Air Resources Board to phase out fumigants that contain SO2F2 because of the chemical’s strong ability to trap heat in the atmosphere.
1. CAS Reg. No. 7783-81-5.
2. CAS Reg. No. 2699-79-8.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Hexa-peri-benzocoronene fast facts
| CAS Reg. No. | 190-24-9 |
| SciFinder nomenclature | Hexabenzo[bc,ef,hi, kl,no,qr] coronene |
| Empirical formula | C42H18 |
| Molar mass | 522.59 g/mol |
| Appearance | Yellow to orange-yellow crystals or powder |
| Melting point | >600 °C |
| Water solubility | Insoluble |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

