The first time Beth Zotter tried her company’s bacon, it tasted bitter, and powdery. “Most protein concentrates don’t taste very well,” says Zotter, cofounder and chief executive officer of Umaro Foods. Umaro was attempting to re-create crispy, savory bacon out of seaweed.
Why bacon? “It’s America’s favorite food,” said co-founder Amanda Stiles on an episode of the TV show, “Shark Tank,” where the two raised funds for Umaro. “It’s the holy grail of plant-based meat. Sizzling, salty, delicious.” But the real magic of Umaro’s pitch was not the bacon. It was the algae.
Plant-based meats are often assembled from soy or pea protein. While algae are plant-like because they conduct photosynthesis with chloroplasts, they don’t have other roots or stems. And they actually photosynthesize far more efficiently than plants.
This holds true from microalgae (or phytoplankton), which are invisible to the naked eye, to enormous forests of seaweed in the ocean.
“Algae own the carbon cycle,” says Schonna Manning, a phycologist (one who studies algae) at Florida International University. “When it comes down to carbon sequestration, algae can sequester up to 400 times more than trees,” said Manning. Algae are an inherently more efficient biological machines for carbon capture than trees or plants, because their entire surface area is dedicated to photosynthesis, and they don’t waste resources creating trunks, roots, or branches. Algae just make more algae.
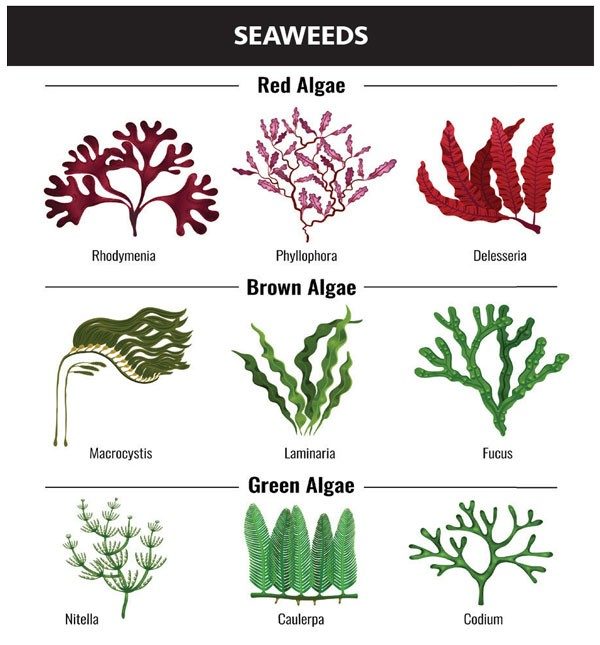
You might think of algae as the dark stuff on your sushi or the layer of green growth inside a forgotten water bottle. But their many species (up to one million) can be harvested and engineered into products that are not just useful, but better than what we use now.
“They’re really little green machines—or purple machines, or golden machines,” says Manning.
These biological machines have the tools to capture planet-warming carbon dioxide from the atmosphere, produce proteins and oils for human nutrition, biofuels, toxins that kill microbes, or miscellaneous vitamins and nutrients that support health.
“There are so many unique groups with all these different unique characteristics that we can exploit if we’re being creative as biologists and chemists and engineers,” said Manning.
Like Umaro, labs around the world have embraced algae as a material for the future. And they are confident that the world will soon depend on seaweed, and be better off as a result.
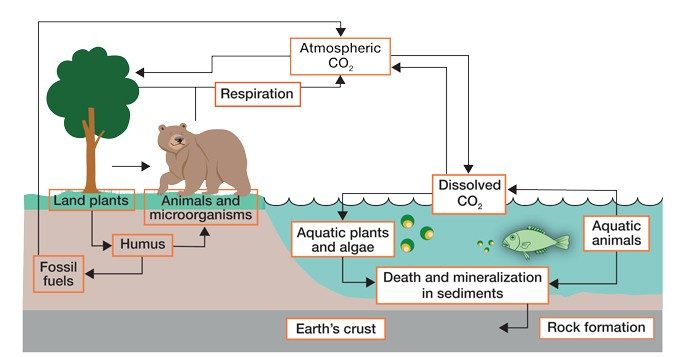
The Carbon Cycle
The carbon cycle is nature’s way of recycling carbon atoms, which move from the atmosphere into living and nonliving things and then back into the atmosphere over and over again.
For example, carbon dioxide is used by plants to make food, the food is eaten by land animals and given back to the atmosphere as they breathe out carbon dioxide.
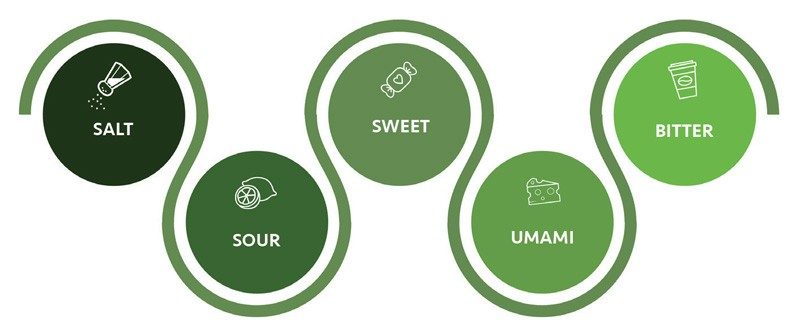
Five Basic Tastes
Biology's First Supermaterial
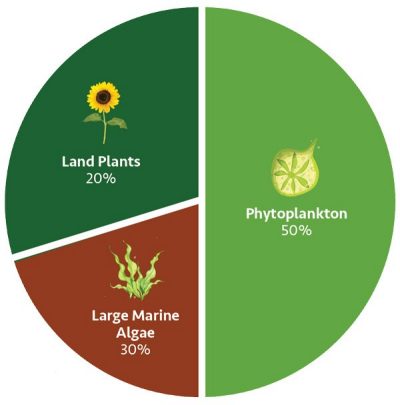
The story of algae’s future promise reflects its ancient past. The oldest algal fossils date back more than one billion years. “Every four out of five breaths that you’re taking in, that oxygen is coming from microalgae not trees,” says Manning. “They’re really a foundation to oxygen and life on this planet.”
Primitive algae were among the first organisms to sequester carbon from the atmosphere, which established their place at the base of the food web.
Brown seaweed also provides us with a compound called alginate. Alginates are copolymers, or chains consisting of two different monomers: β-D-mannuronate (M) and α-L-guluronate (G).
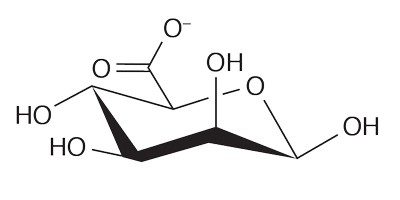
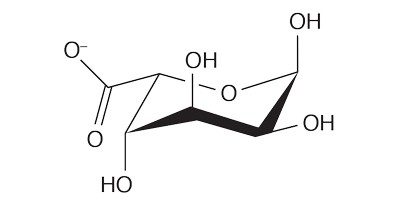
Some sections of that chain feature a sequence of repeating α-L-guluronates, or repeating β-D-mannuronates with some sections alternating between the two.
What results is a gummy, edible polymer. Engineers commonly use alginates in cosmetics, fire-proofing materials, and as a food thickener.
“If you enjoy boba tea, you can thank those engineers because those are actually alginate-de-rived compounds,” says Janine Hutchison, a microbiologist with the Pacific Northwest National Laboratory.
Hutchison and her colleagues develop technologies based on algae and alginate. Last year, they reported creating “3D bio-ink” from alginate. This ink is colorful, lustrous, biocompatible, and can be printed at room temperature, an ideal trait for many applications.
Scientists want to print human tissue in specific three-dimensional shapes. The reason is that real human tissue exists in 3D networks, yet most conventional studies are ducted in 2D Petri dishes.
Cells cultured on three dimensional “scaffolds” behave more like the real thing. It’s better to print human neurons in a small, interconnected sphere than a flat sheet, for example.
Pharmaceutical experiments on these models will better predict actual human responses to new medicines. It’s worth noting that agar and agarose gels used for 2D experiments also come from algae!
“We have been hindered in 3D-printing technologies by relying on plastics,” says Hutchison.
Plastics must be heated to 240 ºC (464 °F) to be able to melt for extrusion.
“There’s no way a living cell, or any biological component can survive that type of heat or stress, even for a short duration of time,” said Hutchison. Alginate is more amenable to biofriendly conditions, so human cells will grow on structures made of alginate.
World Oxygen Production
A Sense for the Future
Algae expands the limits of what people can sense, too. In fact, seaweed is the reason why we discovered a fifth taste.
In 1907, a Japanese woman named Tei Ikeda prepared boiled tofu in a kelp broth called kombu dashi. Dashi had long been a staple of Japanese cooking. But that day, Tei’s husband Kikunae tasted the broth and had a realization: There is a taste here that is distinct from sweet, salty, bitter, and sour.
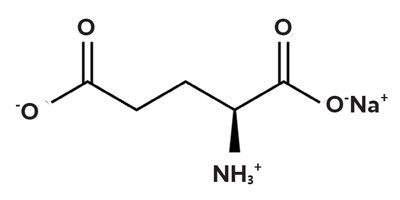
The broth was rich and savory, almost meaty. Kikunae, a chemist, then isolated crystals responsible for the mysterious flavor he named “umami” (translating loosely to “deliciousness”). He discovered that glutamic acid, one of the most common amino acids, was responsible for the taste of umami. He would eventually isolate monosodium glutamate (MSG) which would be sold as a seasoning product for umami.
Umami is now recognized as our fifth taste. It gives us a name to explain why some foods are so good. Ripe tomatoes, egg yolks, aged cheeses such as parmesan, seaweeds such as nori, and cured meats such as bacon, all contain glutamates.
Given this history—and chemistry—it makes sense that Zotter would mimic bacon with seaweed. Meat production accounts for nearly 60% of global greenhouse gas emissions in agriculture, so reducing or replacing it has become a popular approach among people who want to minimize their carbon footprint.
The demand for meat substitutes continues to grow and companies, such as Beyond Meat and Impossible Foods, have led the way with plant-based burgers and ground meats. “Give a lot of credit to the founders of those companies for saying we can make meat replacements from plants,” Zotter says.
But other meats, such as bacon, have been harder to replicate with the usual ingredients. “Right now, we basically have soy, and we have legumes. And some people are now producing using fungi,” she adds.
“We need more options. And the ocean just seems like a no brainer,” said Zotter.
The ocean is an obvious target, according to Zotter, because of nitrogen. Nitrogen is an essential element for life. It’s what gives amino acids their name (from nitrogenous “amine” -NH3+ groups), and snaps them together like Lego pieces to create all proteins.
While gaseous diatomic nitrogen makes up 78% of our air, it’s not chemically reactive nor available for most land plants to process. Oceans contain the largest reservoir of chemically reactive nitrogen on Earth.
“Oceans are huge, and we can make protein without freshwater. The resource potential is enormous,” says Zotter.
Umaro Foods successfully isolated the protein from nori, a protein-rich red algae used on sushi rolls, but bacon is more than just protein. It’s the fat and flavor that sets bacon it apart.
Bacon fat is solid at room temperature, so Zotter’s team found that, when nori protein is combined with algal components agar and carra-geenan it behaves the same as bacon.
Both compounds are hydrocolloids, meaning they form gels with water. They come from gracilaria and kappaphycus species of algae, respectively. These seaweed-based ingredients are able to encapsulate plant-based oils into a fat that crisps and crunches just like the animal fat of bacon. Then, after much trial and error, Umaro Foods developed a flavor recipe to match bacon’s flavor and complement nori’s natural umami.
They debuted their crispy bacon prototype on “Shark Tank,” and now offer it at select restaurants around the United States. Zotter says, “Our vision is that seaweed is the most scalable, lowest cost, and will be the most abundant protein. It’s almost inevitable.”
Re-Greening the World
While algal proteins could reshape the energy in our diets, algal oils could change the fuels that power our machines. Microalgae can reach 50% oil content, by weight, and 25% protein. They are also entirely photosynthetic.
Compare that to a tree that is covered in non-photosynthetic brown bark. This means algae can produce energy efficiently, while simultaneously extracting carbon dioxide from the atmosphere.
Research organizations have wanted to harness algal oils to make fuels, such as jet fuel, for decades, and Dave Hazlebeck, founder of Global Algae, has been a part of this endeavor for nearly 20 years.
Hazlebeck led the biofuels research for an energy company called General Atomics. He steered a consortium of 30 companies in a government-funded program to create jet fuel from algae.
“Through that I became fully aware of all the challenges, all the issues, all the opportunities with algae,” he says. That program ended for the government, but not for Hazlebeck. “I wanted to stay in algae because of the potential of what it could do for the world,” said Hazlebeck.
During the past nine years, Hazlebeck’s Global Algae developed technology to farm and harvest microalgae, and to extract their oils and protein.
They figured out how to cultivate large ponds of microalgae outdoors without contamination from bacteria, viruses, or undesirable algae. Their harvesting system uses 100 times less energy than previous systems.
Each innovation has brought down the cost of producing algae, its oils, and protein, so that it can compete with existing crops such as soy, corn, and palm oil. Existing plant crop farms create problems for the planet—problems that algae may actually solve.
Corn and soy require precious supplies of fresh-water and farmland; the world’s growing reliance on inexpensive palm oil has increased deforestation. Algae farms could replace palm plantations.
And according to Hazlebeck, algal oils are so similar to palm oil, that they can be processed by the same mills. Microalgae are actually richer in omega-3 fats, which could be sold as a separate product.
“You can allow most of the rainforests to regrow, because we can produce almost 10 times as much oil per acre,” Hazlebeck says.
Some of the algae oil would be processed to make jet fuel, and they could isolate the protein from each harvest, too. “About 15 times more than if you were doing soy there,” according to Hazlebeck.
He imagines that protein could feed livestock or fish farms. Each product sets Global Algae up to replace the status quo with alternatives that reduce greenhouse gas emissions. And Hazle-beck also believes algae will help remove carbon dioxide already in the atmosphere.
The idea begins with algae capturing CO2 during photosynthesis. If you make those photosynthetic products into fuel and food, which then get burned or digested, that carbon goes right back into the atmosphere. That’s not a good carbon-se-questration strategy.
But if you turn some of the organic matter from algae into plastics, the carbon will stay trapped for a very long time. Plus, most of the world’s plastic production comes from fossil fuels.
Next, Global Algae plans to use revenue from their products to buy and protect rainforests. Those land buys would just factor into their costs. “It would be an economically viable approach,” Hazlebeck says.
Last year, Global Algae won a $1 million prize by being a finalist in a carbon-capture tech competition led by the nonprofit XPrize. “That was a great honor and helps us by opening doors to move things faster,” says Hazlebeck.
“If we were on an accelerated timeline, we could probably stop deforestation by approximately 2032, and probably have it completely reversed by 2040,” Hazlebeck says.
The final round of demonstrations for XPrize’s carbon-capture contest is in 2025. In the meantime, Global Algae plans to operate a new farm in California and continue refining its extraction technology. The grand prize is $50 million.
Whether you’re interested in inks, plastics, fuel, or food, algae is sneaking into the conversation. The phycologist Manning notes algae’s potential in medicines that combat bacteria, viruses, or even cancer. Many drugs in your medicine cabinet came from plants discovered in rainforests.
Algae’s chemical secrets may also reveal some superpowers. Preliminary studies show that com-pounds isolated from algae can kill drug-resistant pathogens. “Kind of a neo-Neosporin,” says Manning, “Algae don’t have teeth and claws, so they’re producing a lot of these different compounds as a defense. They’re usually just trying to claim their space in the world.”
And algae’s space in the world appears to be on the cusp of a major expansion.
REFERENCES
https://www.xprize.org/prizes/carbon/articles/this-algae-eats-co2-from-the-atmosphere
Yong, E. The Origin Story of Animals Is a Song of Fire and Ice. The Atlantic, Aug 16, 2017: https://www.theatlantic.com/science/archive/2017/08/theanimal-origin-story-is-a-song-of-ice-and-fire/537003/ [accessed Feb 2023].
Zimmer, C. How Did Plants Conquer Land? These Humble Algae Hold Clues. The New York Times, Nov 19, 2019: https://www.nytimes.com/2019/11/14/science/plant-genes-evolution.html [accessed Feb 2023].
Cabrera, J. Innovators Develop Seaweed-Based Alternatives to Plastic Food Wrappers. Mongabay, Jan 17, 2023: https://news.mongabay.com/2023/01/innovators-develop-seaweed-based-alternatives-to-plastic-food-wrappers/ [accessed Feb 2023].
Oster, L. Is Seaweed the Next Big Alternative to Meat? Smithsonian magazine, June 23, 2022: https://www.smithsonianmag.com/innovation/is-seaweedthe-next-big-alternative-to-meat-180980299/ [accessed Feb 2023].