What molecule am I?
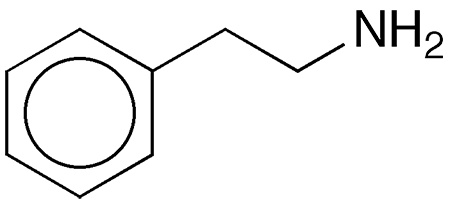
2-Phenylethylamine is a water-soluble amine with a fishy odor. It is often referred to simply as “phenethylamine”, but that can also be interpreted as 1-phenylethylamine1.
2-Phenylethylamine occurs widely in nature: in animals, plants, fungi, and bacteria alike. It has been known since at least 1890; and it was synthesized in 1909 by Treat B. Johnson and Herbert H. Guest at Yale University (New Haven, CT) via the reduction of benzyl cyanide with sodium in ethanol. Later synthetic methods included benzyl cyanide reduction with hydrogen over a Raney nickel catalyst and the reduction of β-nitrostyrene with lithium aluminum hydride.
2-Phenylethylamine is a central nervous system stimulant and is related to many psychoactive compounds such as the amphetamines and catecholamines. Noted (and controversial) chemist/psychopharmacologist Alexander Shulgin performed extensive research on 2-phenylethylamine derivatives, including 3,4-methylenedioxymethamphetamine (MDMA), best known by its street name, “ecstasy”.
Earlier this year, Narciso M. Garrido and colleagues at the University of Salamanca (Spain) published a review of 2-phenylethylamines in medicinal chemistry. Their article focused on open-chain, flexible derivatives such as the catecholamines dopamine, epinephrine (adrenaline), and norepinephrine, compared with constrained polycyclics such as morphine and berberine.
1. CAS Reg. No. 618-36-0.
2-Phenylethylamine hazard information
| Hazard class* | GHS code and hazard statement | |
|---|---|---|
| Flammable liquids, category 4 | H227—Combustible liquid | |
| Corrosive to metals, category 1 | H290—May be corrosive to metals | |
| Acute toxicity, oral, category 3 | H301—Toxic if swallowed | |
| Skin corrosion/irritation, category 1B | H314—Causes severe skin burns and eye damage | |
| Serious eye damage/eye irritation, category 1 | H318—Causes serious eye damage | |
| Short-term (acute) aquatic hazard, category 3 | H402—Harmful to aquatic life | |
*Globally Harmonized System (GHS) of Classification and Labeling of Chemicals. Explanation of pictograms.
MOTW updates
Quercetin1 was the Molecule of the Week for December 9, 2013. It is a plant flavonoid that is used as a supplement to treat various health conditions. The US Food and Drug Administration designates it as “generally recognized as safe” but has not approved it for any medical use. In a report earlier this month, Xingtao Zhou, Shaoping Nie, and co-workers at Nanchang University (China) found that quercetin alleviated liver injury in mice caused by acrylamide, a common contaminant in carbohydrate-rich foods that are processed at high temperatures. The researchers’ analysis showed that quercetin reversed the ferroptosis signaling pathway upregulated by acrylamide.
Zinc oxide2 (ZnO) was the Molecule of the Week for September 22, 2014. It is a widely used inorganic compound, with applications in pigments, ceramics, medicines, and sunscreens. In recent years, it has been used as a wide–band gap semiconductor for optical and electronic applications. This month, Unnikrishnan Manju and co-workers at the CSIR-Institute of Minerals and Materials Technology (Bhubaneswar) and the Academy of Scientific and Innovative Research (Ghaziabad, both in India) described the influence of pressure on the crystallinity, defect density, size, and morphology of ZnO nanoparticles and the effect of these factors on the particles’ optical properties.
Naproxen3 was the Molecule of the Week for May 18, 2009. Its sodium salt is the active ingredient in Aleve and other over-the-counter nonsteroidal anti-inflammatory drugs used to reduce pain, fever, and inflammation. This month, Xiaohong Guan and collaborators at Tongji University and East China Normal University (both in Shanghai) reported an entirely different use for naproxen. They described a method for using the compound in turn-on chemiluminescent probes for quantifying concentrations of the sulfate radical SO4∙– in oxidation processes for wastewater treatment that use the radical anion.
1. CAS Reg. No. 117-39-5.
2. CAS Reg. No. 1314-13-2.
3. CAS Reg. No. 22204-53-1.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
2-Phenylethylamine
fast facts
| CAS Reg. No. | 64-04-0 |
| SciFinder nomenclature | Benzeneethanamine |
| Empirical formula | C8H11N |
| Molar mass | 121.18 g/mol |
| Appearance | Colorless liquid |
| Boiling point | 197–198 °C |
| Water solubility | 63 g/L (25 °C) |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

