What molecule am I?
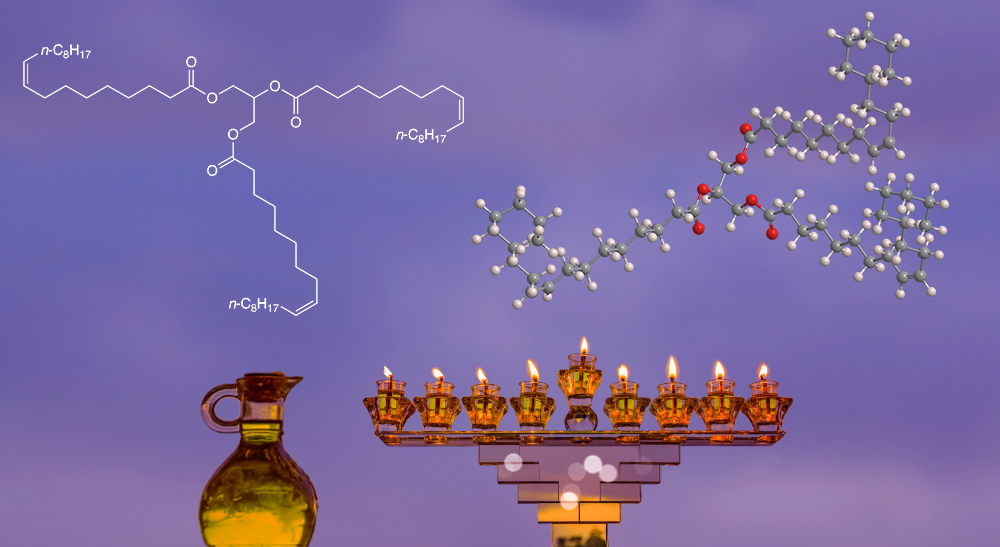
Glyceryl trioleate, known widely as triolein, is an oily liquid that is a main constituent of some nondrying1 oils and fats. Triolein occurs in many natural fats and oils, including sunflower oil, palm oil, cacao butter, and, most notably, olive oil.
In 1941, Thomas P. Hilditch* and L. Maddison at the University of Liverpool (UK) crystallized olive oils from Italy and Palestine at temperatures down to –30 °C to resolve them into several components. They found that olive oil from Palestine contained ≈30% triolein, whereas the Italian oil contained only ≈5%.
Eight years later, M. L. Meara, also at Liverpool, resolved cacao butter into 11 fractions “by exhaustive crystallization”. For his efforts, only 1.1% of the fat turned out to be glyceryl trioleate. From 1940 to 1961, several chemists devised syntheses of triolein by esterifying glycerol and oleic acid.
Why do we celebrate glyceryl trioleate, and therefore olive oil, during the holiday season? Sacramental uses of olive oil are strongly connected to Christian and Jewish traditions, especially Hanukkah.
During the period of the Hanukkah story (168 BCE), only pure olive oil blessed by the high priest could be used to light the Temple menorah, which had to be lit continuously. After their victorious battle over the Syrian Greeks, the Maccabees could find only enough holy oil to last for one day. The Hanukkah miracle is that the oil lasted eight days, enough time for more oil to be prepared and sanctified.
Olive oil was the major component of anointing oils and lamp fuel that date to biblical times. Kings were anointed with oil as a mark of their official status; and one title for Jesus is the Anointed One. References are found throughout the Hebrew and Christian scriptures about the use of oil as part of fasting and healing rituals.
In Imani (faith), the seventh principle of Kwanzaa, designated persons are instructed to pour libations of water and oil into a bowl “for our ancestors; for the dying; for the glorious cloud of witnesses; and for life, health, and strength”.
And if you associate olive oil just with cooking, there are lots and lots of recipes available for cookies and cakes and muffins for your holiday baking!
1. “Nondrying” is somewhat of a misnomer; it means that the substance does not harden when it is exposed to air.
Glyceryl trioleate hazard information
| Hazard class* | GHS code and hazard statement |
|---|---|
| Not a hazardous substance or mixture |
*Globally Harmonized System (GHS) of Classification and Labeling of Chemicals.
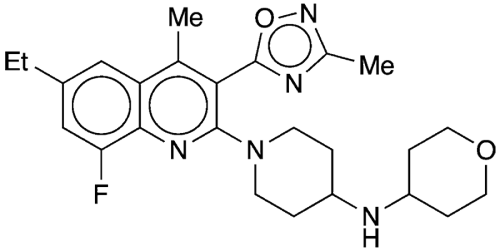
In 2019, Miguel Guerrero, Steven J. Brown, and Edward Roberts at The Scripps Research Institute (La Jolla, CA) and colleagues there and at BlackThorn, the University of Arizona (Tucson), and Scripps (Jupiter, FL) reported that BTRX-335140/CYM-53093, as it was known at the time, is a potent, selective KOR antagonist and that it showed promise for treating a wide range of neuropsychiatric and neurological disorders. Among the potential conditions they said it could ameliorate are migraine, depression, anxiety, drug abuse, and post-traumatic stress disorder.
That same year, biotech startup Neumora Therapeutics was founded with facilities in Watertown, MA, and Brisbane, CA. By October 2021, the company had raised US$400 million in series A financing plus US$100 million from Amgen (Thousand Oaks, CA), with which it also has a research partnership. Neumora also acquired two of BlackThorn’s developmental drugs, including the KOR antagonist, which it renamed NMRA-140. That molecule is in a Phase 2 clinical trial for depression.
1. CAS Reg. No. 2244614-14-8.
Molecule of the Future
Once a month we bring you a newly discovered or developed molecule that has important implications for the future of chemistry or society in general. Look for it the third week of each month. Learn more about this month's Molecule of the Future below.
We're looking for more molecules of the future!
Do you have a suggestion for the next molecule of the future? Send your idea to MOTW.
Glyceryl trioleate fast facts
| CAS Reg. No. | 122-32-7 |
| SciFinder nomenclature | 9-Octadecenoic acid (9Z)-, 1,1’,1″-(1,2,3-propanetriyl) ester |
| Empirical formula | C57H104O6 |
| Molar mass | 885.43 g/mol |
| Appearance | Colorless to yellow oily liquid |
| Boiling point | 235–240 °C (2 kPa) |
| Water solubility | Very slightly soluble |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

