What molecule am I?
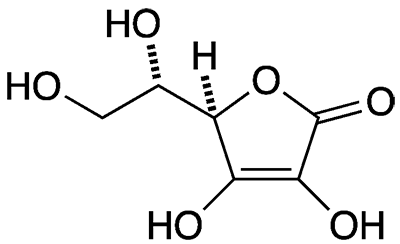
L-Ascorbic acid, more familiarly known as vitamin C, is widely found in nature. It is particularly abundant in citrus and stone fruits, berries (especially acerola), peppers, and green vegetables.
Ironically ascorbic acid was first isolated from an animal source. In 1928, the legendary Hungarian biochemist Albert Szent-Györgyi, then a student at the University of Cambridge (UK), extracted what he called “hexuronic acid” from the adrenal cortices of oxen. He and colleague Walter Norman Haworth later isolated the molecule from lemons and paprika. Szent-Györgyi was awarded the Nobel Prize in Physiology or Medicine in 1937; the same year, Haworth received the prize in chemistry.
In 1933, R. G. Ault at Birmingham Chemical Laboratories (UK), along with Haworth and others, was the first to report a synthesis of L- and D-ascorbic acid. (The D-enantiomer does not exist in nature and has no known uses.) That same year, Polish chemist Tadeusz Reichstein and colleagues at ETH Zürich developed a robust chemical/microbial process for producing ascorbic acid from D-glucose. Their multistep process was the predominant manufacturing route until the 1960s, when it was displaced by a two-step fermentation process in developed in China. Reichstein was awarded the 1950 Nobel Prize in Physiology or Medicine, but for his work on cortisone, not ascorbic acid.
Vitamin C, as ascorbic acid or its salts, is essential for human nutrition. It is a cofactor that permits several enzymes to function properly; it plays a role in the immune system; and it is an antioxidant. Vitamin C deficiency causes the disease scurvy, which, if not treated, results in decreased red blood cell production and excessive bleeding. In extreme cases, it is fatal.
Ascorbic acid, as vitamin C, was the topic of much controversy in the 1970s. Double Nobel prizewinner Linus Pauling advocated the consumption of large doses of the vitamin as a way to compensate for the body’s inability to produce enough of it for optimum health. Specifically, Pauling espoused taking 2.5 g or more vitamin C per day for general health and to prevent or treat the common cold. He and others also contended that doses as great as 10 g/day helped prevent or cure heart disease and cancers. The so-called vitamin C megadose theory has been largely discredited.
Originally the Molecule of the Week for January 1, 2007
Ascorbic acid hazard information
| Hazard class* | Hazard statement |
|---|---|
| Not a hazardous substance or mixture |
*Globally Harmonized System of Classification and Labeling of Chemicals.
Molecule of the future
As the problem of opioid overdosing and addiction continues in the United States and abroad, there is an urgent need to develop alternative, nonaddictive painkillers. Sung Hee Hwang and co-workers at EicOsis LLC (Davis, CA) recently announced the discovery of a new class of analgesics that resolve inflammation and reduce endoplasmic reticulum stress and do not appear to have addictive potential.

The authors found that a specific molecule, called EC5026 (shown), is particularly effective at stabilizing polyunsaturated fatty acid epoxides, which are natural pain mediators. The authors describe the synthesis, optimization, and development of orally administered EC5026 through human Phase 1a trials with no adverse events.
Molecule of the Future
Periodically we bring you a newly discovered or developed molecule that has important implications for the future of chemistry or society in general. Learn more about this week's Molecule of the Future.
We're looking for more molecules of the future!
Do you have a suggestion for the next molecule of the future? Send your idea to MOTW.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule that you would like us to consider, please send us a message, preferably including links to references about your suggested molecule. And thank you for your interest in Molecule of the Week! —Ed.
Ascorbic acid fast facts
| CAS Reg. No. | 50-81-7 |
| SciFinder nomenclature | L-Ascorbic acid |
| Empirical formula | C6H8O6 |
| Molar mass | 176.12 g/mol |
| Appearance | White to pale yellow crystals or powder |
| Melting point | 190–192 ºC (dec) |
| Water solubility | 330 g/L |
MOTW update
Squalene was the Molecule of the Week for December 14, 2020. It is a natural isoprenoid that is an intermediate in the biosynthesis of all animal and plant steroids, including cholesterol. It is most abundant in fish liver oils. In a recent study of ozone pollution, Yingjun Ping at Peking University (Beijing) and international colleagues found that squalene plays a significant role in determining the concentration of ozone inside residences. Squalene is a major component of human skin oils; and the authors found that its reaction products with ozone were abundant in the home that they studied. The concentration of squalene ozonation products correlated with the number of people in the home at any given time.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

